In light of the need for a greater understanding of this nutritional behavior, its effect on an individual’s metabolism and health, and its impact on national health systems, this review was conducted to describe the effect of the Western pattern diet on the metabolism, inflammation, and antioxidant status; the impact on gut microbiota and mitochondrial fitness; the effect cardiovascular health, mental health, and cancer; and the sanitary costs.
However, we have been able to mitigate some of the effects of this discordance through advancements in medicine and technology, which has led to some new challenges given the natural disruption of viral and bacterial processes in modern medicine, such as antibiotic-resistant bacteria [ 15 ]. In addition, the impact of human activities on the environment is creating new selective pressures that could lead to further evolutionary changes in the future. As such, understanding the interplay between genetics and the environment is crucial for developing effective strategies to promote human health and well-being in the face of these challenges.
Indeed, recent clinical trials and interventions have shown that adopting dietary treatments with nutritional characteristics similar to those of pre-agricultural and pre-industrial diets can have positive effects on health [ 14 ]. These findings are consistent with the evolutionary discordance theory, which suggests that the human body is adapted to certain dietary patterns and that modern diets, which differ significantly from those of our ancestors, may contribute to chronic diseases. Thus, by adopting healthier eating habits that are in line with our evolutionary past, we can potentially reduce the risk of chronic diseases and improve overall health.
Thus, measures must be taken to address these issues, such as promoting healthy eating habits and increasing physical activity. Public health campaigns and initiatives can play a crucial role in educating people about the importance of a balanced diet and an active lifestyle [ 13 ]. Policymakers can also implement measures such as food labeling and taxation on unhealthy foods to encourage healthier choices. It is essential to take proactive steps to address these issues and improve the overall health of the nation.
Therefore, there is a dissonance, a change in the food and nutritional model, which reveals a series of epidemiological problems, with an increasing number of many diseases of civilization, including obesity, diabetes, and heart disease [ 7 8 ]. In the United States alone, most adults aged 20 and above, around 65%, are either overweight or obese. This has resulted in an estimated 280,184 deaths every year that can be attributed to obesity alone. Cardiovascular diseases (CVD) are prevalent in over 64 million Americans [ 7 ], and they remain the leading cause of death, accounting for 38.5% of all deaths in the country [ 4 ]. Moreover, 50 million Americans suffer from hypertension and a poor nutritional status, which is a cause of prolonged hospitalization and has a substantial impact on public health cost [ 9 ]. Additionally, 11 million have type 2 diabetes and 37 million have high-risk total cholesterol concentrations of 240 mg/dL [ 9 ]. In postmenopausal women aged 50 years, around 7.2% have osteoporosis and 39.6% have osteopenia [ 10 ]. Additionally, osteoporotic hip fractures have been associated with a 20% excess mortality in the year following the fracture. Cancer is the second leading cause of death in the country, accounting for 25% of all deaths [ 11 ]. An estimated one-third of all cancer deaths are due to poor dietary habits and obesity [ 12 ]. These figures demonstrate the severe impact of a disruptive diet that moves away from the nutritional model of our ancestors [ 1 ], approaching what is known today as the “Western diet”.
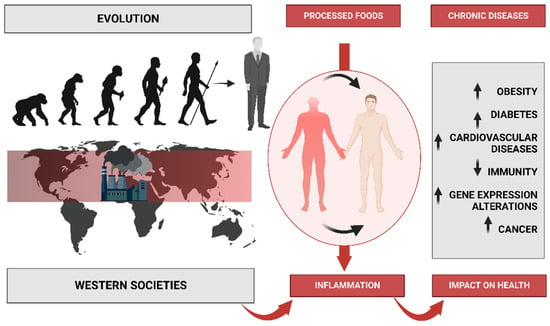
The problem with this is that these changes have occurred too recently on an evolutionary time scale for the human genome to adapt, thus leading to a discordance between our ancient biology and contemporary lifestyle patterns ( Figure 1 ) [ 2 ]. Before agriculture and animal husbandry, hominin diets were limited to minimally processed wild plant and animal foods. However, with the domestication of plants and animals, the nutrient characteristics of these foods changed, which accelerated with advancing technology after the Industrial Revolution [ 3 ]. With the introduction of agriculture, novel foods were introduced as staples for which the hominin genome had little evolutionary experience. Furthermore, food processing procedures were developed that allowed for combinations of nutrients and foods not previously encountered in hominin evolution [ 1 ]. It is crucial to consider not only the nutrient qualities and the types of foods that would have been consumed by preagricultural hominins but also the types of foods and nutrient qualities that could not have been regularly consumed before the development of agriculture, industrialization, and advanced technology. Additionally, it is worth noting that dairy products, cereals, refined sugars, refined vegetable oils, and alcohol make up 72.1% of the total daily energy consumed by all people in the United States [ 4 ], but these types of foods would have contributed little or none of the energy in the typical preagricultural hominin diet [ 5 ]. Additionally, processed foods also dominate in a typical Western diet, such as cookies, cake, bakery foods, breakfast cereals, and snack foods [ 6 ].
Like all species, contemporary humans are genetically adapted to the environment of their ancestors, which conditioned their genetic profile. In terms of nutrition, the introduction of agriculture and animal husbandry about 10,000 years ago has lead to profound changes in the diet and lifestyle of these “modern humans” [ 1 ].
In this investigation, we performed a comprehensive examination of primary and secondary sources that incorporated scientific articles, bibliographic indexes, and databases such as PubMed, Scopus, Embase, Science Direct, Sports Discuss, ResearchGate, and the Web of Science. We applied MeSH-compliant keywords such as Western diet, inflammation, metabolic health, metabolic fitness, cardiovascular disease, cancer, oxidative stress, mental health, and metabolism to explore articles that were published between 1 January 2003 and 1 March 2023. The following exclusion criteria were used: (i) studies with inappropriate or irrelevant topics not pertinent to the main focus of the review, (ii) Ph.D. dissertations, conference proceedings, and unpublished studies. A team of five review authors meticulously evaluated the titles and abstracts of all collected manuscripts to ascertain their suitability. Studies that utilized outdated data, had irrelevant topics that did not align with the research objectives, or were not in English were eliminated. The same team of five review authors who undertook the study selection independently extracted pertinent data from the selected studies. Subsequently, the outcomes were discussed to create the current comprehensive review. It is worth mentioning that this study’s approach guarantees that the included data are current, relevant, and of high quality, thereby making the findings of this review reliable and useful for future research.
The Western diet shifts the co-expression of 445 gene pairs in mice, including small RNAs and transcription factors associated with metabolism and adiposity in humans, and dramatically alters behavior. For example, Western-fed individuals were more anxious and less socially integrated [ 33 ]. Similarly, the Western diet limits microbial short-chain fatty acid (SCFA) production in humans, which prevents many of the microbiota-dependent events from occurring, such as histone deacetylation and DNA methylation, and leads to alterations in hepatic gene expression [ 34 ]. The Western diet evokes structural and behavioral changes in the resident microbiome, which alters the representation of metabolic pathways in the microbiome and alters microbiome gene expression [ 35 ], as well as inducing epigenetic changes in monocytes that affect their inflammatory response and cytokine production [ 33 ], altering the expression of genes involved in lipid metabolism and oxidative stress in liver tissue [ 36 ], and modulating the expression of genes related to the circadian rhythm and melatonin synthesis in the pineal gland [ 37 ]. Similarly, it can also increase gene expression alterations related to a fatty liver (non-alcoholic fatty liver disease).
The Western diet also can influence gene expression and epigenetics. Gene expression refers to the process by which genetic information is translated into functional molecules such as proteins or RNA. Epigenetics refers to the study of heritable changes in gene expression that do not involve changes in the DNA sequence but rather modifications in DNA methylation, histone modification, or microRNA regulation. The Western diet can affect gene expression and epigenetics by altering nutrient availability, cofactor supply, hormonal levels, environmental cues, cellular signaling pathways, transcription factors, chromatin structure, DNA methylation, histone modification, microRNA regulation, feedback loops, genetic variants, gene–environment interactions, intergenerational effects, and transgenerational effects ( Figure 1 ) [ 32 ].
The availability of processed and ultra-processed foods including sugar, industrial seed oils, and poultry has increased over the past two centuries, while butter/lard/shortening, dairy (primarily whole-fat), fresh fruits, fresh vegetables, and red meat (beef/pork) have decreased. Before 1900, ultra-processed foods were uncommon, but they now make up more than half of the American diet [ 28 ]. The gut microbiome is the collection of microorganisms that reside in the digestive tract, including bacteria, viruses, fungi, and other microbes. This complex ecosystem plays an important role in the health of the human body, contributing to various functions such as digestion, immune system regulation, and the synthesis of certain vitamins and other essential nutrients [ 29 ]. In the scientific literature, it has been previously stated that one of the main factors, together with physical exercise, that modulates the intestinal microbiota is nutrition [ 30 ]. However, due to this, diet impacts the gut microbiome and immune system. Concretely, the Western diet can disrupt the balance and diversity of the gut microbiome leading to dysbiosis, which is a condition characterized by an overgrowth of harmful bacteria and a reduction in beneficial bacteria. Dysbiosis can impair intestinal barrier function, increase intestinal permeability, promote bacterial translocation, and trigger systemic inflammation. Dysbiosis can also affect immune system function, modulate immune cell differentiation, alter immune cell activation, influence cytokine production, regulate immunoglobulin secretion, and modulate immune tolerance [ 31 ].
Cardiovascular disease is the leading cause of death worldwide, and the Western diet is a major risk factor for this disease. High intakes of saturated and trans fats, added sugars, and salt have been linked to an increased risk of cardiovascular disease [ 26 ]. In contrast, diets high in fruits, vegetables, whole grains, and healthy fats such as those found in the Mediterranean diet have been shown to reduce the risk of cardiovascular disease [ 16 ]. Certain cancers, particularly colorectal cancer, have also been associated with the Western diet. High intakes of red and processed meats, as well as low intakes of fruits, vegetables, and whole grains, have been linked to an increased risk of colorectal cancer [ 27 ].
The Western diet includes low intakes of fruits, vegetables, whole grains, and nuts. These foods are important sources of vitamins, minerals, fiber, and antioxidants, which are essential for optimal health. Low intakes of these foods have been linked to an increased risk of chronic diseases such as obesity, type 2 diabetes, and cardiovascular disease [ 24 ]. The Western diet has been linked to a range of chronic diseases and inflammation processes ( Figure 1 ), including obesity, type 2 diabetes, cardiovascular disease, and certain cancers. In the United States, the data reveal alarming trends between 1999 and 2018, with male obesity increasing from 27.5% to 43.0% and severe obesity from 3.1% to 6.0%. In women, the prevalence of obesity rose from 33.4% to 41.9%, and the prevalence of severe obesity rose from 6.2% to 11.5% [ 25 ]. Obesity is a major risk factor for a range of chronic diseases, including type 2 diabetes, cardiovascular disease, and certain cancers. The consumption of a Western-style diet has been shown to increase the risk of obesity and related diseases [ 17 ]. Type 2 diabetes is another major health concern associated with the Western diet. A high intake of processed and refined foods, added sugars, and saturated and trans fats has been linked to an increased risk of type 2 diabetes [ 19 ]. In addition, low intakes of fruits, vegetables, whole grains, and nuts have been associated with an increased risk of type 2 diabetes [ 24 ].
In order to clarify which countries are exposed to this diet concept, we provide official FAO data. FAO-provided food balance sheets are adjusted for consumer waste and used to calculate a Western dietary similarity index (WSI) for each nation. Based on this information, a ratio of calories from animal foods, oils, lipids, and sweeteners to total calories per capita can be calculated [ 21 23 ]. A plausible value is WSI = 70, which corresponds to the US WSI in 2013, indicating that the global steady-state WSI will correspond to a dietary pattern in which 70% of calories originate from animal foods, oils and fats, and sweeteners. Iceland (72), Switzerland (72), the United States (70), Australia (69), Sweden (67), Hungary (66), France (66), Austria (66), Germany (66), Denmark (66), the Czech Republic (65), the Netherlands (65), Spain (65), Belgium (65), Finland (64), and New Zealand comprise the “Western diet countries” group (64).
The Western diet is also characterized by high consumption of red and processed meats. Red meat consumption has been associated with an increased risk of colorectal cancer, while processed meat consumption has been linked to an increased risk of colorectal cancer, cardiovascular disease, and type 2 diabetes [ 18 ]. Another major characteristic of the Western diet is the high intake of added sugars. Added sugars are sugars and syrups that are added to foods during processing, such as high-fructose corn syrup in soft drinks and table sugar in baked goods. High intake of added sugars has been linked to an increased risk of obesity, type 2 diabetes, and cardiovascular disease [ 19 ]. The Western diet is also characterized by high intakes of saturated and trans fats. Saturated fats are found in animal products such as meat, butter, and cheese, while trans fats are found in processed foods such as baked goods and fried foods. High intake of saturated and trans fats has been linked to an increased risk of cardiovascular disease [ 20 ].
The Western diet is characterized by a high intake of energy-dense, nutrient-poor foods such as fast foods, soft drinks, and highly processed foods, which are high in added sugars, salt, and saturated fats. In contrast, the traditional diets of non-Western countries, such as the Mediterranean diet, are characterized by high intakes of fruits, vegetables, whole grains, legumes, and healthy fats such as olive oil and nuts [ 16 ]. The consumption of processed and refined foods is a major characteristic of the Western diet. Processed foods are foods that have been altered from their natural state, typically to increase their shelf life or improve their taste. Examples include fast foods, packaged snacks, and sugary drinks. Refined foods are foods that have had their natural fiber and nutrients removed during processing, such as white flour, white rice, and added sugars [ 17 ].
The Western diet has been the subject of much discussion in the field of nutrition. It is a modern dietary pattern that is characterized by high intakes of processed and refined foods, red and processed meats, added sugars, and saturated and trans fats and low intakes of fruits, vegetables, whole grains, and nuts ( Figure 1 ). The Western diet has been linked to a range of chronic diseases, including obesity, type 2 diabetes, cardiovascular disease, and certain cancers. In this paper, we will discuss the nutritional characteristics of the Western diet, its impact on health, and the potential solutions to improve dietary patterns in Western societies [ 12 ].
The food environment, including the availability and accessibility of healthy food options, also plays a role in shaping dietary choices. People living in areas with limited access to healthy food options are more likely to have poor dietary habits. A study by Stowers et al. (2020) [ 44 ] found that individuals living in food deserts had lower intakes of fruits and vegetables and higher intakes of sugar-sweetened beverages compared to those living in areas with better food access. The food environment is also associated with health outcomes related to the Western diet. People living in food deserts are more likely to have poorer health outcomes due to limited access to healthy food options. A study by Caspi et al. (2012) [ 45 ] found that individuals living in food deserts had higher risks for obesity and type 2 diabetes compared to those living in areas with better food access. Understanding these social characteristics is essential for developing effective interventions aimed at promoting healthier dietary habits and reducing the risk of chronic diseases.
Similarly, social norms can also influence dietary choices. People are often influenced by the behavior of those around them, including family, friends, and peers. One study [ 42 ] found that social norms related to healthy eating were positively associated with fruit and vegetable intake among adolescents. Social norms related to healthy eating are associated with better health outcomes. Individuals who perceive social norms to support healthy eating behaviors are more likely to have healthier diets and lower risks for chronic diseases, and adolescents who perceived social norms to support healthy eating had higher intakes of fruits and vegetables and lower intakes of sugar-sweetened beverages [ 43 ].
In addition to the Western diet, sedentary lifestyles and low levels of physical activity are also associated with an increased risk of chronic diseases. In a paper that studied 3316 Finnish participants between 25 and 74 years old, it was found that higher levels of physical activity were associated with a reduced risk of type 2 diabetes [ 40 ]. Similarly, a study with 88 140 participants aged 40–85 years old found that higher levels of physical activity were associated with a reduced risk of cardiovascular disease [ 41 ].
The Western diet has become a global phenomenon and has had significant impacts on human health. Social characteristics such as income, education, and cultural background play a crucial role in shaping dietary habits and preferences. There are some social characteristics of the Western diet, which are related to income and education, since income and education are essential social factors that influence dietary choices. People with higher incomes and more education are more likely to have healthier diets that include more fruits, vegetables, whole grains, and lean proteins. A study by Burrows et al. (2017) found that higher levels of education were associated with a higher intake of fruits and vegetables [ 38 ]. Income and education levels are associated with health outcomes related to the Western diet. Lower-income individuals and those with less education are more likely to have poor dietary habits and be at higher risk for chronic diseases such as obesity, type 2 diabetes, and cardiovascular disease ( Figure 1 ). For example, it was found that low-income individuals were more likely to have a higher intake of processed foods and a lower intake of fruits and vegetables [ 39 ].
In conclusion, the Western diet is associated with a lower antioxidant status compared to healthy dietary patterns, and this may increase the risk of chronic diseases. A diet rich in fruits, vegetables, whole grains, nuts, and legumes can provide essential nutrients, including antioxidants, that are critical for optimal health. Therefore, it is recommended that individuals adopt a healthy dietary pattern that emphasizes plant-based foods and limits the intake of processed and high-fat foods to promote good health and reduce the risk of chronic diseases.
In addition to consuming a diet rich in antioxidants, other lifestyle factors can impact antioxidant status. Physical activity has been shown to increase antioxidant capacity in the body, reduce oxidative stress, and improve overall health [ 84 ]. It was found that individuals who engaged in regular physical activity had higher levels of antioxidant enzymes, such as superoxide dismutase and glutathione peroxidase, compared to sedentary individuals [ 85 ]. Additionally, smoking and alcohol consumption have been associated with decreased antioxidant levels in the body and increased risk of chronic diseases [ 86 87 ].
In a study conducted on overweight and obese adults, the consumption of a Western diet for eight weeks was found to reduce the activity of superoxide dismutase and increase oxidative stress markers in the body [ 82 ]. These findings suggest that a Western diet may increase the risk of chronic diseases by promoting oxidative stress and reducing the body’s ability to defend against it. Low levels of antioxidants can have significant implications for health. Oxidative stress, which results from an imbalance between free radicals and antioxidants in the body, can damage cells, proteins, and DNA. This damage can lead to chronic diseases such as cancer, cardiovascular disease, and neurodegenerative diseases [ 83 ]. In addition, low levels of antioxidants have been associated with an increased risk of inflammation and oxidative damage to the body’s tissues [ 76 ].
Moreover, the Western diet may exacerbate oxidative stress in the body by reducing the activity of endogenous antioxidants such as glutathione and superoxide dismutase ( Figure 2 ) [ 72 ]. Oxidative stress occurs when the body’s antioxidant defenses are overwhelmed by the production of reactive oxygen species (ROS) and reactive nitrogen species (RNS). These free radicals can damage DNA, proteins, and lipids, leading to cell dysfunction and death [ 73 75 ]. Oxidative stress has been implicated in the pathogenesis of numerous diseases, including cancer, neurodegenerative disorders, and cardiovascular disease [ 76 ]. The Western diet is a major contributor to oxidative stress [ 73 75 ]. High intake of processed foods and sugar-sweetened beverages has been linked to increased production of ROS and RNS ( Figure 2 ) [ 10 ]. These foods are often high in refined carbohydrates and saturated and trans fats, which can promote inflammation and increase the production of ROS and RNS [ 77 ]. Red and processed meat, another hallmark of the Western diet, are also associated with oxidative stress. The heme iron in red meat can catalyze the formation of ROS, leading to oxidative damage [ 78 ]. Additionally, processed meats contain nitrates and nitrites, which can react with other compounds in the body to form RNS [ 79 ]. Refined grains, another staple of the Western diet, are low in fiber and other important nutrients, which can increase inflammation and oxidative stress [ 80 ]. High intake of refined grains has been associated with increased levels of markers of oxidative stress in the blood [ 81 ].
Studies have found that the Western diet is associated with lower antioxidant status in the body, as measured by levels of antioxidant enzymes and biomarkers [ 68 ]. This may contribute to the development of chronic diseases, as low antioxidant status has been linked to increased oxidative stress and inflammation [ 69 ]. Therefore, improving antioxidant status through dietary changes may have potential health benefits, particularly in individuals with a high intake of the Western diet. One study found that increasing the consumption of fruits and vegetables, which are high in antioxidants, was associated with a lower risk of mortality from cardiovascular disease and cancer [ 70 ]. Another study found that supplementation with antioxidant vitamins reduced the risk of cardiovascular events in high-risk individuals [ 71 ].
Several studies have highlighted the importance of a diet rich in antioxidants for maintaining optimal health ( Figure 2 ) [ 61 ]. A high intake of fruits and vegetables, which are rich sources of antioxidants, has been associated with a reduced risk of chronic diseases, [ 62 ] including cardiovascular disease, cancer, and neurodegenerative disorders [ 63 64 ]. In contrast, a Western diet that is low in fruits, vegetables, and whole grains and high in saturated fats, sugar, and processed foods has been linked to a higher risk of chronic diseases. A study conducted in the United States found that individuals consuming a Western diet had a higher risk of heart disease, diabetes, and stroke than those consuming a healthy diet [ 65 ]. Furthermore, the Western diet not only lacks essential antioxidants but also contains pro-oxidant compounds that increase the production of free radicals and oxidative stress in the body [ 66 ]. A study conducted on a sample of healthy adults found that consuming a high-fat meal induced oxidative stress, increased inflammation, and decreased antioxidant capacity in the body [ 67 ]. The study also found that consuming a meal rich in fruits and vegetables before the high-fat meal reduced oxidative stress and inflammation and improved antioxidant capacity.
Since the diet type analyzed in this paper is deficient in antioxidants, the problem is exacerbated [ 54 55 ].The main sources of dietary antioxidants are fruits, vegetables, whole grains, and nuts. However, the Western diet is deficient in these foods, which means that individuals following this dietary pattern are more likely to have low levels of antioxidants [ 56 ]. Studies have shown that individuals following the Western diet have lower levels of the previously mentioned vitamin C, vitamin E, and beta-carotene than those consuming a healthy diet [ 57 58 ]. For example, a study conducted on a sample of adults in the United States found that those following the Western diet had lower serum levels of vitamin C and vitamin E than those following a healthy diet [ 59 ]. Similarly, a study conducted on Australian adults found that those consuming a Western diet had lower serum levels of beta-carotene than those consuming a healthy diet [ 60 ].
The Western dietary pattern has been associated with an increased risk of chronic diseases, and one of the mechanisms by which the Western diet may contribute to the development of these diseases is through oxidative stress [ 46 ]. Oxidative stress occurs when there is an imbalance between the production of reactive oxygen species (ROS) and the body’s antioxidant defense system ( Figure 2 ). In this process, a free radical can be defined as any molecular species capable of independent existence that contains an unpaired electron in an atomic orbital. The presence of an unpaired electron gives rise to certain common properties that most radicals share. Many radicals are unstable and highly reactive. They can either donate or accept an electron from other molecules, so they behave as oxidizers or reductants [ 47 ]. As a consequence of an imbalance between free radical production and antioxidant defenses, oxidative stress is associated with injury to a broad spectrum of molecular species, including lipids, proteins, and nucleic acids [ 48 ]. Due to important macromolecules being attacked by free radicals, cell injury and a disruption of homeostasis develop [ 49 ]. An antioxidant is a molecule that is stable enough to donate an electron to a free radical and neutralize it, thereby reducing its destructive potential [ 50 ]. These antioxidants delay or prevent cellular injury primarily due to their ability to neutralize free radicals. These low-molecular-weight antioxidants can interact with free radicals and terminate the chain reaction without causing injury to vital molecules. Some of these antioxidants, such as glutathione, ubiquinol, and uric acid, are produced by the body’s normal metabolism [ 51 ]. There are additional milder antioxidants in the diet. Although there are numerous enzyme systems in the body that neutralize free radicals, the most important micronutrient (vitamin) antioxidants are vitamin E (tocopherol), vitamin C (ascorbic acid), and beta-carotene. Because the body cannot produce these micronutrients, they must be consumed [ 52 ]. In summary, antioxidants can neutralize ROS and prevent oxidative damage. However, the Western diet is generally low in these antioxidant-rich foods and high in pro-oxidant substances such as omega-6 fatty acids and advanced glycation end products (AGEs) [ 53 ].
Regarding gut diseases, dysbiosis, or an imbalance of the intestinal microbiota, has been linked to the Western diet and may contribute to inflammation. Dysbiosis of the gut microbiota describes a state of discord between the microbes normally found in the digestive tract and their host. The Western diet lacks the fiber and prebiotics necessary to foster the development and variety of good gut flora [ 109 ]. Instead, the diet is high in fat, sugar, and salt, which can promote the growth of harmful bacteria and fungi. This can result in gut inflammation, increased gut permeability, and the release of bacterial endotoxins, such as lipopolysaccharides (LPS), into the bloodstream. These endotoxins can activate the immune system and promote the release of pro-inflammatory cytokines, such as IL-1β and IL-18 [ 110 ]. Previous authors found that the Western diet was associated with decreased gut microbial diversity and increased levels of pro-inflammatory bacteria, such as Proteobacteria [ 111 ]. The dysbiosis induced by the Western diet can lead to increased intestinal permeability, allowing bacterial endotoxins to enter the bloodstream and trigger inflammation [ 112 ].
To continue, it is relevant to note that the method of cooking is also thought to alter or may affect metabolic processes. In relation to this, advanced glycation end products (AGEs)—compounds created when food is cooked at very high temperatures—are abundant in the Western diet. When AGEs bind to the RAGE receptor, pro-inflammatory cytokines are released [ 103 ]. AGEs are a group of complex molecules formed by the non-enzymatic reaction between reducing sugars and amino acids or proteins. This reaction is known as the Maillard reaction and occurs naturally in the body as part of normal metabolism, but also through external sources such as cooking methods, particularly high-temperature cooking, and the processing of food [ 104 ]. AGEs are known to accumulate in tissues over time and contribute to the development of various diseases [ 105 106 ]. AGEs are formed through the non-enzymatic reaction between reducing sugars and amino acids and are present in high levels in processed foods, particularly those that are high in fat and sugar [ 103 ]. The Western diet is high in AGEs due to the consumption of processed and refined foods, which are often cooked at high temperatures, leading to the formation of AGEs [ 107 ]. These AGEs can be found in various foods, including meat, dairy, and baked goods, which are staple items in the Western diet [ 103 ]. It was found that participants who consumed a high-AGE diet for four weeks had increased markers of inflammation and oxidative stress, which are known to contribute to the development of chronic diseases such as diabetes and cardiovascular diseases [ 108 ]. AGEs can activate the receptor for advanced glycation end products (RAGE), which is expressed on the surface of many cell types, including endothelial cells, macrophages, and adipocytes. RAGE activation leads to the production of pro-inflammatory cytokines and oxidative stress, which are involved in the pathogenesis of chronic diseases [ 103 ].
Given the preceding, all of the evidence indicates that high-fat diets result in immune dysregulation. Specifically, low-grade inflammation is promoted by an imbalance in the actions of the immune system. In the Western diet, sugar and fatty acids can either be pro-inflammatory—such as omega-6—and present in high numbers, or they can be anti-inflammatory—such as omega-3 or antioxidants—and present in lesser proportions [ 99 ]. This can promote the activation of immune cells, such as macrophages and T cells, and the release of pro-inflammatory cytokines, such as IL-1β, IL-6, and TNF-α [ 100 ]. The consumption of high levels of saturated and trans fats found in the Western diet can activate Toll-like receptors (TLRs) on immune cells, leading to the production of pro-inflammatory cytokines and chemokines [ 101 ]. TLRs are important mediators of inflammatory pathways that detect pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs) and initiate an immune response [ 102 ]. In response to a Western diet, the TLR pathway is activated by the high levels of endotoxins and other pro-inflammatory molecules present in the diet. This activation leads to the production of pro-inflammatory cytokines, such as TNF-α, IL-6, and interleukin-1 beta (IL-1β), which are involved in the pathogenesis of chronic diseases [ 101 ].
In addition to providing evidence along these lines, rodent studies may be pertinent to dietary changes in humans. Concretely, recent studies found that the consumption of a high-fat diet led to increased production of ROS in rats [ 96 ]. It was also shown that antioxidants lose their efficacy in an inflammatory state, making this a progressive, hard-to-control rise in oxidative stress [ 97 ]. These findings suggest that the consumption of a Western diet may contribute to the development of chronic diseases through increased ROS production. Similarly, processed and fried foods, baked goods, and high-fat dairy products have been shown to increase LDL cholesterol levels and contribute to the development of cardiovascular diseases [ 98 ]. Thus, high-fat diets are particularly harmful, as they not only increase LDL cholesterol levels but also decrease HDL cholesterol levels, leading to an unfavorable lipid profile [ 98 ].
Multiple mechanisms, including oxidative stress, intestinal microbiota dysbiosis, and immune dysregulation, have been also shown to contribute to the pro-inflammatory effects of a Western diet ( Figure 1 ). When there is an imbalance between the production of reactive oxygen species (ROS) and the body’s ability to detoxify, oxidative stress occurs [ 74 93 ]. Thus, saturated and trans fats, which can increase ROS production and impair antioxidant defenses, are the main promoters of oxidative stress creation [ 73 ]. This can result in damage to cells, tissues, and organs, and the release of pro-inflammatory cytokines, such as IL-6, and TNF-α [ 94 ]. ROS are highly reactive molecules that are formed as a byproduct of cellular metabolism, and their production is tightly regulated in the body. However, oxidative stress conditions injure cells and tissues [ 95 ]. This damage can contribute to the development of chronic diseases such as cardiovascular diseases, diabetes, and cancer [ 95 ].
High consumption of saturated fats, processed foods, and refined sugars is characteristic of the Western diet and has been associated with persistent low-grade inflammation. Critical in the body’s defense against infections and injuries, inflammation is a complicated process involving the immune system [ 88 ]. Specifically, chronic inflammation can lead to the development of several chronic diseases, such as cardiovascular disease, type 2 diabetes, and cancer ( Figure 1 ) [ 89 90 ]. In this regard, research has demonstrated that Western diet patterns, which are also associated with a high intake of red and processed meats, refined cereals, and caffeinated beverages, promote elevated levels of-reactive protein (CRP), a well-established marker of inflammation according to previous research [ 91 ]. Additionally, authors have also reported that the Western diet was associated with increased levels of CRP and other inflammatory markers, such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) [ 92 ]. In this case, the WD results in a persistent metabolic inflammatory state [ 66 ].
Although the gut microbiota can be widely utilized as a tool for food safety assessment, more research is needed to better understand the relationship between the gut microbiota and food consumption. While there is mounting evidence for the potential link between gut microbiota and health outcomes, further studies are necessary to establish a causal relationship.
In contrast, vegetarian or plant-based diets may be beneficial to the gut microbiome [ 141 ]. Dietary polyphenols are plant-based natural chemicals found in vegetables, grains, fruits, coffee, and tea [ 142 ]. These organic molecules are a complex and heterogeneous class of substances characterized by hydroxylated phenyl groups, which are present in very small concentrations in the Western diet [ 143 ]. They are frequently divided into flavonoids and non-flavonoids according to their chemical structure and complexity. The influence of dietary polyphenols on gut ecology and the mechanisms behind the effects on intestinal and extraintestinal illnesses has recently been explored. Various research works have discovered antioxidant, antidiabetic, anticarcinogenic, neuroprotective, anti-inflammatory, cardioprotective, antibacterial, antiadipogenic, and other properties in various dietary polyphenols [ 144 ].Concretely, epidemiological studies have found links between the consumption of polyphenol-rich foods such as vegetables and drinks and illness prevention. These results have been seen in certain types of cancer, cardiovascular disease, type 2 diabetes, osteoporosis, pancreatitis, gastrointestinal problems, lung damage and neurodegenerative diseases [ 145 ].
One of the groups of foods that seem to have a greater impact on the intestinal microbiota and that receive the most attention in the scientific literature are ultra-processed foods [ 132 ]. These comprise food products made from substances extracted from whole foods that are then combined with various additives, such as preservatives, flavorings, and sweeteners [ 133 ]. Ultra-processed foods, such as soft drinks and savory snacks, are highly appealing due to their long shelf-life, affordability, and convenience for consumption anytime and anywhere [ 134 ]. Their consumption is alarmingly high in many high-income countries. For instance, in France [ 135 ], ultra-processed food contributes to 29.1% of total energy intake, while in Australia and the United States, it accounts for 42% and 57.9% [ 136 ], respectively. The negative effects on the gut microbiota [ 6 ] have been studied by authors such as Zinöcker MK and Lindseth IA. Authors suggest that ultra-processed foods that contain high levels of added sugars, unhealthy fats, and sodium alter the gut microbiota, leading to a reduction in beneficial bacteria and an increase in harmful bacteria. Moreover, these foods are low in fiber, which is a critical nutrient that promotes the growth of beneficial gut bacteria. In this regard, a recent meta-analysis concluded that vegetable consumption was associated with a lower risk of ulcerative colitis alone, whereas fruit consumption was associated with a lower risk of both Crohn’s disease (CD) and ulcerative colitis [ 137 ]. However, authors also suggest that the emulsifiers and additives used in the production of ultra-processed food may have harmful effects on gut microbiota, including inflammation and increased permeability of the gut lining. Additionally, some studies suggest that the consumption of ultra-processed foods may lead to changes in brain activity and behavior, including addiction-like behaviors, which can contribute to overconsumption and further health problems [ 138 ]. Analysis of the feces of adults consuming aspartame and acesulfame K in a recent cross-sectional clinical study with 31 participants did not reveal an increase in bacterial abundance, but rather a decrease in bacterial diversity compared to non-consumers. As mentioned, the data may be inconsistent; the median Bacteroidetes:Firmicutes ratio did not change in seven participants who consumed 1.7–33.2 mg/day of acesulfame K for four days, but bacterial diversity statistically differed from that of non-consumers [ 139 140 ].
Dysbiosis and the process of chronic inflammation, as well as the profile and nutritional quality of these typical Western foods, occur simultaneously. Intestine dysbiosis is a condition that occurs when the natural equilibrium of microorganisms in the intestine is disturbed, resulting in an overgrowth of harmful bacteria or a reduction in beneficial bacteria [ 127 ]. The foods we consume provide not only the essential nutrients required for our health but also create an environment for the growth and sustenance of gut bacteria. The composition of our diet plays a significant role in determining the type of bacteria that flourish in our gut. As the importance of gut microbiota in maintaining good health has been recognized, there has been growing interest in studying the impact of diet on gut microbiota [ 111 130 ]. Specifically, the Western diet model is hypothesized to increase proinflammatory cytokines, disrupt the epithelial barrier, and alter the intestinal microbiota (with an increase in Bacteroides and Enterobacteriaceae and a decrease in Bifidobacterium and Lactobacillus), thereby promoting low-grade chronic inflammation in the gut [ 131 ].
Similarly, authors have suggested that the Western diet promotes inflammation in the body by increasing the levels of pro-inflammatory molecules such as cytokines and-reactive protein [ 122 ]. Furthermore, the intake of saturated and trans fats, as well as refined carbohydrates, typical in this diet may trigger the release of inflammatory mediators [ 123 ]. In contrast, a Mediterranean diet, rich in fruits and vegetables, whole grains, and lean protein sources such as fish, can help reduce inflammation and protect against chronic diseases [ 124 ]. Illescas et al. found in a meta-analysis that diets and gastrointestinal maladies are linked to inflammation and cancer, highlighting the distinctive characteristics of the bacterial population associated with the Mediterranean diet. In particular, the microbiota of subjects following a MD was enriched with pro-anti-inflammatory bacteria. For the first time, studies revealed an increase in Akkermansia and a decrease in Fusobacterium with a MD, even below the levels observed in healthy individuals with no defined diet without a diet plan. Fusobacterium is a known pathogenic bacterium associated with malignancy, whereas Akkermansia is indicative of a healthy gastrointestinal tract [ 125 ]. One of the main reasons for this is the anti-inflammatory capacity provided by the antioxidants in this diet. These foods are high in antioxidants, which can neutralize free radicals and prevent oxidative damage to cells. Additionally, they contain anti-inflammatory compounds, such as polyphenols and omega-3 fatty acids, which can help reduce inflammation in the body [ 126 ].
In addition, the combination of a high intake of fat and sugar with a low intake of fiber, fruits, and vegetables is associated with chronic inflammation. Consequently, inflammation is an important underlying mechanism connecting the Western diet to chronic diseases [ 120 ]. Regarding this, Bolte et al. demonstrated through long-term diet protocol research that higher abundances of Firmicutes, Ruminococcus species of the Blautia genus, and endotoxin synthesis pathways were consistently associated with processed and animal-derived diets. In contrast, plant and fish diets were found to be positively associated with short-chain fatty acid-producing commensals and nutrient metabolism pathways [ 121 ].
Based on the above, our knowledge reveals that eating a lot of processed, high-calorie meals is not only associated with an increased risk of developing conditions including diabetes, obesity, heart disease, but also gut dysbiosis. Concretely, the high meat consumption related to the Western diet increases Bacteroides, Alistipes, and Bilophila (associated with pathological processes such as atherogenesis) and decreases Bifidobacterium, Roseburia, Eubacterium, and Ruminococcus [ 113 ]. Additionally, the Firmicutes/Bacteroidetes ratio has been frequently considered a plausible marker for obesity over the past decade and a parallel of Western diet patterns [ 114 ]. In this regard, the abundance of Firmicutes in the gastrointestinal microbiota of healthy individuals ranges from 11% to 95%, while that of Bacteroidetes ranges from 0.6% to 86.5%. However, Kasai et al. reported more accurately that Firmicutes comprised 37.0 ± 9.1% of non-obese and 40.8 ± 15.0% of obese individuals, whereas Bacteroidetes comprised 44.0 ± 9.8% of non-obese and 37.0 ± 14.0% of obese individuals [ 115 ]. These alterations in bacterial composition and/or diversity are typically associated with alterations in the microbiota’s metabolic profile, which also impact host health [ 116 ]. However, the relative abundance of the phyla Firmicutes and Bacteroidetes is highly variable among members of the same population [ 117 ]. This is likely due to the influence of numerous harmful lifestyle factors, such as the Western diet [ 118 ], low physical activity, dietary additives and contaminants, or antibiotic consumption. This may explain the contradictory results observed when comparing the microbiota of normal-weight and obese subjects, making it challenging to correlate the ratio of Firmicutes to Bacteroidetes with a particular health status [ 117 ]. Although the intestinal microbiota may contribute to the development of obesity, there is insufficient evidence to support a link between obesity and changes in the Firmicutes/Bacteroidetes ratio [ 119 ].
Regarding mitophagy during HFD, it has been found that in healthy, non-obese, sedentary males and 10 endurance-trained male runners, the protein content of total PTEN-induced kinase 1 (PINK1) in skeletal muscle was found to be similar between groups before and after a high-fat meal, indicating that mitophagy is not required for metabolic flexibility in the healthy population [ 163 ]. However, it has been demonstrated that mice fed an HFD (60 kcal% fat) for two months display mitophagy, which is followed by decreased mitochondrial abundance (mtDNA/nuDNA) [ 164 ]. Additionally, a decrease in PARKIN in mitochondria that were supposed to be recruited by PINK1 has been demonstrated [ 165 ]. Although mitochondria can readily adapt to exogenous parameters and maintain their proper functionality to generate sufficient energy for cellular demands, these organelles are sensitized to variations in substrate availability due to different dietary habits, followed by molecular rewiring. This orchestrates alterations in mitochondrial function, regulation of biogenesis and mitophagy, and ‘self-recycling’ via fusion and fission in mitochondria. In conclusion, a high-fat diet impairs mitochondrial function and fusion while inducing fission.
Although an HFD has been shown to lead to insulin resistance, impaired mitochondrial function, and increased production of reactive oxygen species (ROS), much more needs to be known about the dysregulated expression profile of proteins involved in mitochondrial dynamics. Regarding the proteins involved in mitochondrial dynamics, an HFD diet caused mitochondrial fusion protein 2 (MFN2) to drop and proteins involved in fission processes to rise (Drp1 and FIS1) [ 159 ]. In this regard, Chen et al. also confirmed that male Sprague Dawley rats fed an HFD (60 percent kcal) showed increased mitochondrial FIS1 levels [ 16 ]. Consistent with this, it was also found that healthy male C57BL/6 mice that were 4 weeks old and fed an HFD for 16 weeks showed reduced relative expression of MFN2, but higher levels of mitochondrial DRP1 expression [ 160 ]. Lionetti et al., according to electron microscopy and immune activity analysis, suggested that this type of diet also caused a shift towards fission in skeletal muscle. These results are consistent with earlier reports that type 2 diabetic patients and obese Zucker rats have decreased MFN2 expression in their skeletal muscles [ 159 ]. Furthermore, in the work of Jheng et al., differentiated C2C12 skeletal muscle cells exposed to saturated fatty acids have been shown to trigger fission processes in vitro that are linked to mitochondrial dysfunction [ 161 ]. In addition, high-glucose (HG) treatment has been linked to mitochondrial fragmentation and an increase in fission processes in myoblasts and a rat liver cell line. It has been hypothesized that this fragmentation would increase the total surface area of metabolically active organelles, increasing the accessibility of metabolic substrate to carrier proteins. As a result, it is possible to hypothesize that HFD-induced mitochondrial fragmentation is a cellular adaptation to increase mitochondrial intake and oxidize excess dietary fatty acids, which would lead to increased ROS production [ 159 162 ].
The traditional Western diet (WD) pattern’s dietary traits are specifically a high intake of saturated fats and omega-6 fatty acids, a decreased intake of omega-3 fats, an abundance of processed carbohydrates, and an overuse of salt [ 8 ]. Drinks account for 47% of the added sugars in this surplus, which amounts to more than 13% of the daily calorie intake [ 7 ]. Thus, considering the WD as a high-fat diet (HFD), one of the most important pathophysiological symptoms of HFD is insulin resistance, which causes obesity, among other diseases [ 7 ]. One of the well-explained pathophysiological changes that an HFD promotes is impaired mitochondrial biogenesis ( Figure 3 ). Concretely, excessive calorie consumption causes tissue hypertrophy and adipocyte hyperplasia in white adipose tissue [ 152 ]. The subsequent manifestations increase lipolysis in fat cells and ultimately result in raised amounts of free fatty acids (FA) in circulation. Enhanced lipid catabolism and energy production via the Krebs cycle have already been established as outcomes of increased FA oxidation in the mitochondria [ 152 ]. The adipocyte-secreted hormones adiponectin, leptin, acylation-stimulating protein, and resistin play a crucial role in regulating mitochondrial biogenesis and insulin sensitivity [ 153 154 ]. In this regard, adiponectin physically binds to its receptors during HFD intake, activating AMP-activated protein kinase (AMPK), which ultimately stimulates glucose uptake and FA oxidation. Moreover, AMPK has been linked to the control of PGC-1α, the principal regulator of mitochondrial biogenesis [ 155 ]. According to studies on animals, PGC1 α mRNA and protein levels in skeletal muscle were likewise decreased in C57Bl/6 mice fed an HFD for three weeks [ 156 ]. Diastolic dysfunction and poor mitochondrial function were associated in a different investigation. Nevertheless, PGC-1 and mitochondrial transcription factor A (TFAM) protein levels were elevated in the gastrocnemius muscle of male and female Wistar rats that were 2 months old and fed an HFD for 26 weeks. The same study hypothesized that as male rats showed a larger rise in PGC-1 α and TFAM levels than female rats, the effects of an HFD on mitochondrial biogenesis may be sex-dependent [ 154 ]. As previously mentioned, a prolonged HFD affects the availability of substrates that mitochondria can use; as a result, mitochondria need to adapt their molecular machinery to generate ATP effectively and sufficiently, especially in high-energy consuming tissues, such as the brain and myocardium. In HFD-fed (60% kcal) rats, Chen et al. reported that reduced complex I-III and citrate synthase activities have been found, which are significant findings that support compromised cardiac function. Similarly, rats showed decreased mitochondrial respiration activities [ 157 ], which also supports compromised cardiac function. Likewise, it has been demonstrated that feeding male Wistar rats an HFD for 4 weeks increases their levels of OXPHOS-related proteins such as cytochrome c oxidase (COX)I, COXIV, uncoupling protein (UCP) 3, and cytochrome b [ 158 ]. Despite the fact that some studies have shown an increase in the OXPHOS complex, perhaps as a compensatory mechanism to enable mitochondria to produce more energy, an HFD appears to impair mitochondrial function, affecting cellular energetics and ATP turnover [ 152 ].
Individuals’ diets consist of mixtures of foods and beverages (referred to as foods for simplicity). However, the precise combination of foods that constitutes a healthy diet is context-dependent and influenced by several cultural, economic, and other variables [ 146 ]. In recent decades, global food habits have shifted towards increasingly Westernized and less healthy diets. Based on the World Health Organization’s (WHO) worldwide nutrition report, while a portion of the world’s population suffers from starvation, the other portion suffers from obesity and its accompanying problems [ 147 ]. With the right proportion, content, quantity, and presence of macronutrients, micronutrients, and bioactive chemicals, a balanced diet counteracts these severe situations. Unfortunately, little is understood about how these components affect human health. These nutrients destined to nourish our bodies, tissues, and cells must first reach the mitochondria, where they are broken down into COand HO to produce energy. Mitochondria are the cell’s powerhouse and are primarily responsible for nutrition metabolism [ 148 ]. Nevertheless, they are also the primary cause of oxidative stress and apoptosis-mediated cell death [ 149 ]. However, mitochondrial performance might be “improved” by physical activity, which improves their integrity, efficiency, and dynamic adaptability to stressors, thus, “mitochondrial fitness” [ 150 ]. Mitochondria are extremely dynamic organelles, and their bioenergetics are directly linked to the fusion/fission equilibrium. Fusion activities relate to the enhancement of mitochondrial function, whereas fission processes are associated with the elimination of dysfunctional mitochondria. These processes are influenced by the consumption of a variety of foods, which gives cells several stimuli. Concretely, they are influenced by the energy substrate availability caused by food quality and diet timing. In this regard, it is well acknowledged that different dietary fat sources have distinct effects on the dynamics and bioenergetics of the mitochondria, including mitophagy ( Figure 3 ) [ 6 ]. For instance, obesity-related disorders such as insulin resistance and non-alcoholic fatty liver disease are greatly influenced by mitochondrial dysfunction [ 151 ].
Implementing various strategies that resulted in a marked decrease in inflammation diseases and consequently CVD, with an enormous increase in life expectancy as a result, was one of public health’s greatest achievements. However, over the past few decades, more and more populations have been exposed to a Western lifestyle, which is associated with a high consumption of processed foods high in calories, which has resulted in an increase in diseases that are largely preventable and linked to a chronic inflammatory state [ 7 ]. In fact, we face a pandemic of diseases linked to lifestyle because more than one third of adults worldwide are considered overweight or obese. As society becomes increasingly health-conscious, healthy dietary patterns may increase. In the upcoming years, formal dietary recommendations by major health organizations will ultimately be guided by newly emerging data highlighting new definitions of a healthy diet and which foods make up this diet. In the coming decades, the current trend towards healthier choices may extend life expectancy and improve cardiovascular outcomes [ 172 209 ].
Conversely, the published literature generally does not support statistically significant associations between dietary cholesterol and CVD risk. The heterogeneity in the adjustment for total energy, other dietary components, and serum cholesterol concentrations shows that studies of dietary cholesterol have been conducted over a long time span during which nutritional epidemiology methods have significantly changed. Nonetheless, it has been demonstrated by Zhong et al. that there is a positive correlation between dietary cholesterol and egg consumption and the risk of CVD. According to the authors’ calculations, the risk of CVD increased significantly with every additional half egg consumed each day. However, when dietary cholesterol was taken into account, the link between egg consumption and CVD was eliminated [ 201 ]. Table 1 provides further references for all of the above and specifically for the benefits of plant-based or vegetarian diets.
Well-established knowledge suggests that Western dietary patterns, in comparison to healthier dietary patterns such as the “Mediterranean diet” (MeDiet), are associated with an increase in the production of proinflammatory cytokines and a decrease in the production of anti-inflammatory cytokines [ 188 190 ]. Therefore, greater adherence to healthier eating habits, which include consuming more fruits, vegetables, legumes, nuts, and whole grains, may lessen low-grade inflammation and thereby prevent cardiovascular disease [ 191 192 ]. A cohort study carried out by Gao et al. identified food-based dietary patterns that operate through excess energy intake and explained high variability in energy density, free sugars, saturated fat, and fiber intakes to investigate their association with total and fatal CVD and all-cause mortality [ 193 ]. High intakes of chocolate and sweets [ 194 195 ], butter, and low-fiber bread and low intakes of fresh fruits and vegetables comprised the predominant dietary pattern and were positively associated with total CVD [ 193 ]. In contrast, current evidence demonstrates that healthy dietary patterns are characterized by a high intake of fiber, antioxidants, vitamins, minerals, polyphenols, and monounsaturated and polyunsaturated fatty acids and a low intake of salt, refined sugar, saturated and trans fats, and carbohydrates with a low glycemic load [ 99 196 ]. This corresponds to a high intake of fruits, vegetables, legumes, fish and seafood, nuts, seeds, whole grains, vegetable oils (primarily extra virgin olive oil), and dairy foods, and a low intake of sweets, soft drinks, and red and processed meat [ 197 198 ]. Concretely, in a recent meta-analysis, which included 86 cross-sectional and 10 cohort prospective studies for a total population of more than 130,000 vegetarians and 15,000 vegans (plant-based diet, complete abstention from meat and meat products, poultry, seafood, and consumption of any other product from an animal), it was found that vegetarians and vegans had significantly lower levels of BMI, total cholesterol, LDL-cholesterol, and glucose than omnivores. Additionally, an analysis of prospective studies showed a 25% pooled significant risk reduction in ischemic heart disease incidence and/or mortality [ 199 200 ].
Current scientific evidence demonstrates that chronic inflammation plays an important role in the pathogenesis of coronary artery disease (CAD), including the initiation and progression of atheroma plaque and rupture, as well as post-angioplasty and restenosis.-reactive protein (CRP), interleukin (IL)-1, IL-6, IL-8, IL-1, IL-18, monocyte chemoattractant protein (MCP)-1, and tumor necrosis factor (TNF α), among others, are the primary mediators of CAD development. Additionally, the results of Usui et al.´s study show that IL-17 is crucial for the emergence of moderate atherosclerosis and shed new light on IL-17’s function in the pathogenesis of atherosclerosis [ 184 ]. Moreover, these mediators are considered potential biomarkers of inflammation, and their expression may be correlated with the severity of coronary artery disease [ 185 186 ]. Oikonomou et al. provided further evidence assessing 118 stable symptomatic patients and concluded that the Western diet (increased intake of fat, red meat, and carbohydrates and low consumption of fruits and green leafy vegetables) was predictive of severe CAD [ 187 ].
A variety of conditions affecting the heart and blood vessels are referred to as CVD, including hypertension, strokes, atherosclerosis, peripheral artery disease, and vein diseases [ 174 ]. The combination of unhealthy nutritional habits and habits typical of Western societies creates an environment that is highly prone to cardiovascular system failure ( Figure 4 ) [ 175 176 ]. Concretely, ASCVD is an inflammatory disease that contributes significantly to the incidence and mortality of CVD. Libby, on the one side [ 177 ], and Mozzaffarian et al. [ 178 ], on the other, pointed out in a study based on atherosclerosis therapies that oxidative stress and systemic inflammation are modifiable by nutrition; however, excessive energy intake and physical inactivity contribute to the secretion of pro-inflammatory cytokines [ 179 ]. Inflammatory processes involve the subendothelial region of the arterial wall, where lipids and macrophages loaded with lipids accumulate [ 180 ]. Concretely, Virmani et al. specified that early stages of atherosclerosis are characterized by the internalization of lipids in the intima, primarily low-density lipoproteins (LDL), which results in endothelial dysfunction. Endothelial dysfunction disruption promotes inflammatory process, emboli, and multiple pathological outcomes, along with calcifications, stenosis, burst, and hemorrhage. Advanced atherosclerosis manifests clinically as coronary heart disease, ischemic stroke, peripheral artery disease, heart failure, or sudden death [ 181 182 ]. However, Yubero-Serrano’s findings demonstrate that the Mediterranean diet, especially in CHD patients with severe endothelial dysfunction, controls endothelial function better than a low-fat diet and is linked with a better balance of vascular homeostasis [ 183 ].
In Western societies, cardiovascular disease (CVD) is the leading cause of death, with coronary heart disease (CHD) accounting for more than 50% of cases. Numerous risk factors, including family history, diabetes, hypertension, obesity, smoking, and inactivity, contribute significantly to the overall cardiovascular risk ( Figure 4 ). Recent data indicate an interesting gradient in the incidence, morbidity, and mortality of cardiovascular disease across the socioeconomic status spectrum, as defined by educational level, occupation, or income [ 166 ]. Personal characteristics, such as physical inactivity, smoking, stress, and alcohol consumption, only account for 13.6% of the SE differences in CVD morbidity and 19.5% of the SE differences in CVD mortality ( Figure 4 ) [ 167 ]. Despite the above-mentioned established facts, dietary interventions are much less frequently used in the management of cardiovascular disease than pharmacological and procedural interventions. Nutritional guidelines recommend eating a diet high in fish [ 168 ], whole grains [ 169 ], vegetables and fruits [ 170 ], legumes [ 171 ], and nuts to reduce the risk of developing atherosclerotic cardiovascular disease (ASCVD). To lower the risk of ASCVD, dietary monounsaturated and polyunsaturated fats should be substituted for saturated fat, along with sodium, cholesterol, processed meats, refined carbohydrates, and sweetened beverages [ 172 ].the consumption of Western-type diets (WDs), which exclude fiber, vitamins, and minerals, has increased over the past few decades in Westernized societies. WDs include processed foods, “fast food”, convenience foods, snacks, and sugary soft drinks ( Figure 4 ). With the spread of these food items and their consumption from high- to low-income countries, there has also been a parallel rise in diseases, as mentioned for CVD, linked to the Western diet [ 77 173 ].
After reviewing the Western diet, it is possible to affirm that its effects on general health, and more specifically on mental health, are not beneficial. The food composition of this diet, rich in processed foods, red meat, and saturated fats among others, as well as the lack of fiber and vitamins, makes the body vulnerable to the presence of mental pathologies. Among the most important are mood disorders in the face of chronic low levels of inflammation, which appear as a defense and repair mechanism in the face of intestinal dys