As such, we propose the repurposing of existing LDH inhibitors to impedegrowth. Here, we evaluate the effects of commercially available LDH inhibitors on the in vitro growth ofusing a standardized growth assay. The LDH inhibitors that we used cover a variety of mechanisms: competitive in respect to pyruvate (oxamate), competitive in respect to NADH (gossypol, AT-101, GSK2837808A), competitive with both NADH and pyruvate (NHI-2, galloflavin, FX-11), and noncompetitive inhibitors (stiripentol, isosafrole) [ 10 25 ]. To a lesser extent, gossypol also shows noncompetitive inhibition of pyruvate [ 26 ]. The understanding of these mechanisms, in conjunction with in vitro performance, will guide future in vivo evaluations of these compounds and, ultimately, the development of a new generation of tools for managing Lyme disease.
LDH inhibitors have been widely suggested for the treatment of various cancers and apicomplexan infections [ 8 11 ]. In humans, there are a multitude of LDH types consisting of different tissue-associated combinations of two subunits, with the most prominent combinations being classified as LDHA (skeletal LDH) and LDHB (heart LDH) [ 12 ]. Both LDHA and LDHB play roles in the Warburg effect, the phenomenon of increased levels of anaerobic glycolysis observed in cancer cells compared to noncancerous cells [ 12 13 ]. Under these conditions, LDH becomes an indispensable resource to cells and its inhibition is shown to reduce tumor growth [ 14 ]. Compared to current anticancer chemotherapies, selective LDH inhibition has few side effects: complete LDHA deficiency in humans is almost universally asymptomatic, further increasing the attractiveness of anti-LDH drugs for cancer chemotherapy [ 15 ]. Importantly, commercially available LDH inhibitors show efficacy against the LDH of eukaryotic microorganisms such as those possessed by 9 ], as well as those for several species of tickborne pathogens within the genus 17 ].
Lyme disease’s causative agent,, has a highly reduced genome and is therefore relegated to a specialized metabolism.lacks a tricarboxylic (TCA) cycle, cannot conduct oxidative phosphorylation, and has no pathways for carbohydrate, amino acid, or lipid biosynthesis [ 6 ]. Instead, glycolysis is the sole mechanism for’s ATP production. Furthermore, because it lacks both pyruvate dehydrogenase and pyruvate oxygenase, the sole means by whichcan use pyruvate is by conversion to lactate via the enzyme, lactate dehydrogenase (LDH) [ 7 ]. Thus, LDH is an essential metabolic linchpin for, and its inhibition could serve as an effective method for mitigating cell growth. This atypical metabolic circumstance presents an opportunity to target the spirochete with minimal collateral effects on the host.
Lyme disease is the most common zoonotic illness reported in North America. The range of the disease’s primary arthropod vectoris expanding, potentially increasing the risk of infection in much of the United States [ 1 ]. Current treatment strategies are effective in most cases, but as many as 17% of patients may remain partially symptomatic one year after treatment [ 2 ]. Chemoprophylactic doxycycline dosing has been shown to effectively reduce the risk of Lyme disease manifestation following tick bite exposure [ 3 ]. However, prophylaxis may be problematic due to potentially adverse side effects of antibiotic treatment, the relatively low rate of disease manifestation after single tick bites, and the general risks associated with antibiotic overuse [ 4 5 ]. As such, expanding the arsenal of existing Lyme disease management tools is undoubtedly beneficial.
Analyses were conducted using GraphPad Prism 8.3.2 for Windows (GraphPad Software, San Diego, CA, USA. To account for variations in cell growth between replicate experiments, growth numbers were normalized within each experiment, compiled, and fitted with a nonlinear regression line. Normalization was relative to the average cell growth of each experiment’s vehicle control group, with the control quantified as 100% and instances with no growth (≤100 visible cells) quantified as 0%. IC 50 values were determined using dose-dependent normalized response curves.
Our MBC determination methodology was adapted from Sicklinger et al. [ 28 ]. In instances where no bacterial growth was evident, a 25 μL sample was taken from all replicates with inhibitor concentrations greater than or equal to the MIC. Each sample was added to 1 mL of inhibitor-free BSK-H. Cells were then incubated for two weeks at 34 °C, after which they were observed for living spirochetes under a darkfield microscope. If no motile spirochetes were visible, the concentration of the LDH inhibitor was considered bactericidal. The reported MBCs are the mode values at which concentrations were considered bactericidal.
Each drug concentration was tested in triplicate. In cases where strong dose-dependent responses were present, experiments were repeated with alterations to drug dosage to better determine the half-maximal inhibitory concentration (IC 50 ). The minimum inhibitory concentration (MIC) was defined as the smallest dosage required to fully inhibit bacterial growth and was calculated as the midpoint between the lowest bacteriostatic concentration and the highest non-bacteriostatic concentration. For example, in a case where no cell growth was seen at a concentration of 100 uM of an LDH inhibitor and the next highest dosage was 50 μM of a given inhibitor, we would estimate the MIC as 75 μM.
cells were grown from the previously created cryovials, as described above.was diluted into a large single stock of 50 mL BSK-H and mixed thoroughly to ensure a homogeneous distribution of cells. Cells were taken from this working mixture and added to inhibitor–BSK solutions, achieving a final concentration of 1.0 × 10cells/mL. Following the method of Caol et al. [ 27 ], cells were then incubated at 34 °C for 72 h, before being counted manually using darkfield microscopy. Bacteriostatic concentrations were defined as any concentration after 72 h in which cell counts did not exceed the base concentration. To allow for random fluctuations in cell counts, three dilutions to 10cells/mL were conducted, and each was counted three times. The standard deviation was calculated. Assuming a normal distribution of cell counts, twice this standard deviation was added to the initial inoculum, establishing 1.33 × 10cells/mL as the cutoff point for bacteriostatic concentrations.
LDH inhibitors were dissolved in DMSO, added to BSK-H complete media, and then sterilized using vacuum filtration (0.22 μm PVDF filter). Oxamate is not sufficiently soluble in DMSO and was instead directly dissolved into BSK-H complete media before filtration. In-house vehicle control data indicate no significant difference ingrowth in unaltered media compared to media with concentrations of less than 0.5% DMSO ( Figure S1 = 0.94). LDH inhibitors were then serially diluted in BSK-H media to achieve the desired final concentrations.
Our growth inhibition methodology was adapted from Caol et al. [ 27 ]. A uniformstock was created and used for all assays.strain B31 clone 5A2 was acquired from BEI Resources (Manassas, VA, USA) as a frozen glycerol stock. A low-passage (<3) sample was incubated in closed, screw-cap vials containing 14 mL BSK-H complete media (Sigma Aldrich) at 34 °C for nine days, until cells reached a concentration of greater than 10cells/mL. Cells were manually counted utilizing darkfield microscopy and C-Chip disposable hemocytometers (INCYTO), before being diluted to 1.1 × 10cells/mL in 50 mL of BSK-H complete media. Next, 900 μL aliquots were added to cryovials, and 100 μL DMSO was added as cryoprotectant. Cells were stored at −80 °C.
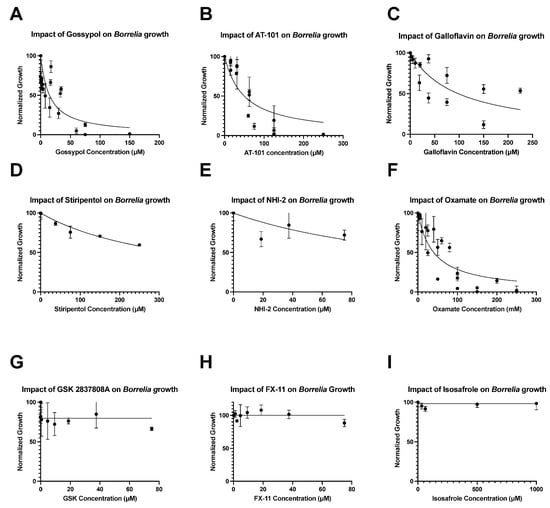
The inhibitors, isosafrole, FX-11, and GSK2837808A, exhibited little to no effect on Borrelia growth in vitro ( Figure 1 G–I). Therefore, no MIC, MBC, or ICcould be determined for these compounds, and no significant inhibitory effect was seen, even at the highest concentrations of the inhibitors ( Table 1 ).
Galloflavin and stiripentol also showed strong dose-dependent effects ongrowth, with ICs of 98 μM and 350 μM, respectively, but did not yield MIC or MBC values at the tested concentrations ( Figure 1 C,D). Similarly, while NHI-2 had a noticeable impact ongrowth at high concentrations with an ICof 150 μM, the overall dose dependence was low: data were highly variant, and the growth curve had an Rvalue of only 0.32 ( Figure 1 E).
The impacts of LDH inhibitors ongrowth varied greatly between compounds ( Table 1 ). Of all compounds, racemic gossypol was shown to have the lowest ICand MIC values: 14 μM and 70.5 μM, respectively. Notably, the levorotatory enantiomer of gossypol, AT-101, demonstrated a significantly lower impact than the racemic mixture with ICand MIC values of 50 μM and 187.5 μM, respectively ( Figure S2 = 0.0026). AT-101 and racemic gossypol both demonstrated bactericidal properties: AT-101 was found to have an MBC value of 250 μM, while gossypol displayed an MBC value of 150 μM.
4. Discussion
Borrelia burgdorferi . As such, a range of commercially available LDH inhibitors with various mechanisms of action and origins were tested on Borrelia in culture. Of these inhibitors, gossypol, AT-101, and oxamate substantially impacted B. burgdorferi growth in vitro and represent promising candidates against Borrelia infections in vivo. While oxamate’s IC 50 concentration is easy to achieve in vitro due to its high solubility in water, it requires a dose several orders of magnitude higher than other active compounds. Nonetheless, it should be noted that these concentrations are comparable to those found for the treatment of human cancer cells [22, LDH inhibitors have been tested against cancer cells and apicomplexans but are less well studied against bacteria [ 8 10 ]. Furthermore, this is the first instance that LDH inhibitors have been tested on. As such, a range of commercially available LDH inhibitors with various mechanisms of action and origins were tested onin culture. Of these inhibitors, gossypol, AT-101, and oxamate substantially impactedgrowth in vitro and represent promising candidates againstinfections in vivo. While oxamate’s ICconcentration is easy to achieve in vitro due to its high solubility in water, it requires a dose several orders of magnitude higher than other active compounds. Nonetheless, it should be noted that these concentrations are comparable to those found for the treatment of human cancer cells [ 29 ]. As such, it may be possible that oxamate is more effective when used in tandem with other compounds, rather than as a sole treatment option, as is demonstrated in multiple anti-cancer studies [ 8 30 ].
dextrorotatory gossypol more effectively inhibits human placental LDH [ Borrelia growth inhibition. This differential is promising for dextrorotatory gossypol’s potential as a treatment option: gossypol toxicity is primarily caused by the levorotatory enantiomer [ In terms of bacteriostatic inhibition, racemic gossypol was shown to have a lower MIC than AT-101, the levorotatory enantiomer of gossypol. Prior studies have shown the variance in the efficacy of the gossypol enantiomers, even when used against human LDH subtypes: levorotatory gossypol more effectively inhibits human LDHA [ 31 ], whilegossypol more effectively inhibits human placental LDH [ 32 ]. The increased efficacy of racemic gossypol relative to AT-101 suggests that dextrorotatory gossypol is the more effective enantiomer ingrowth inhibition. This differential is promising for dextrorotatory gossypol’s potential as a treatment option: gossypol toxicity is primarily caused by the levorotatory enantiomer [ 24 33 ]. Likewise, though gossypol has been tested as a male infertility agent, levorotatory gossypol is the primary cause of antifertility activity [ 34 ]. Thus, focusing on the dextrorotatory enantiomer may significantly reduce concerns of both toxicity and infertility.
Borrelia cells. Isosafrole’s exact mechanism and site of interaction remains unknown, and the relatively large size (MW = 649.6) and branching topology of NADH-competitive GSK2837808A likely compromises its scope in targeting LDH types from evolutionarily distant organisms [ B. burgdorferi LDH ( Bb LDH) modulators. FX-11 and gossypol are expected to differ in charge under physiological conditions, since the former is a carboxylic acid; however, protein sequence alignment attests to the high conservation of charged residues in the vicinity of the substrate-binding site in both human LDHA and Bb LDH ( Borrelia activity altogether is counterintuitive, since the latter is expected to fit where the larger counterpart binds. Although both FX-11 and GSK2837808A are shown to be effective on human cancer cells in vitro at nanomolar and micromolar levels [ 10 25 ], and isosafrole is shown to be a potent human LDHA inhibitor [ 22 ], these agents had no effect oncells. Isosafrole’s exact mechanism and site of interaction remains unknown, and the relatively large size (MW = 649.6) and branching topology of NADH-competitive GSK2837808A likely compromises its scope in targeting LDH types from evolutionarily distant organisms [ 35 ]. However, the lack of activity of gossypol-like FX-11 is more intriguing and may offer some insight into the further design of potentLDH (LDH) modulators. FX-11 and gossypol are expected to differ in charge under physiological conditions, since the former is a carboxylic acid; however, protein sequence alignment attests to the high conservation of charged residues in the vicinity of the substrate-binding site in both human LDHA andLDH ( Figure S3 ). Hence, this lack of activity cannot be readily attributed to a difference in charge but instead points to size as the characteristic that causes the highly different activities of these structurally related compounds. The fact that racemic gossypol is the most active of all tested agents, while its smaller variant FX-11 lacks anti-activity altogether is counterintuitive, since the latter is expected to fit where the larger counterpart binds.
Bb LDH and relies on its bulk to reach the level of affinity that translates into observed spirochetal growth suppression, whereas smaller substrate-competitive inhibitors target the closed form of LDHA. The minimal activities of such inhibitors against B. burgdorferi suggest that mobile loop closure may amplify differences between rather conserved active sites in human LDHA and Bb LDH, pointing to the open conformation for development of effective chemotherapeutics against Borrelia . These differences are also reflected in the observation that the two enzymes display distinct preferences for the two enantiomers of gossypol. While human LDHA is more sensitive to its levorotatory form, Bb LDH apparently is inhibited more effectively by dextrorotatory gossypol. These observations will guide modeling studies to develop more effective anti- Borrelia chemotherapeutics. The disparity between gossypol and FX-11 efficacy may be explained by the well-established structural dynamics of the active site in LDH enzymes. The LDH active site mobile loop can adapt to either open and closed states depending on the substrate and cofactor association status [ 36 ]. Unlike the more compact inhibitors, FX-11 and galloflavin, which are competitive with both pyruvate and NADH [ 37 ], bulky and rigid gossypol does not fit into the compact pocket formed upon mobile loop closure and is therefore expected to bind to the ‘open’ forms of LDH ( Figure S4 ). Gossypol is only competitive with NADH and non-competitive with pyruvate [ 26 ], suggesting that it targets a conformational state where the substrate subsite is not yet fully organized. Consequently, we hypothesize that gossypol associates with an open form ofLDH and relies on its bulk to reach the level of affinity that translates into observed spirochetal growth suppression, whereas smaller substrate-competitive inhibitors target the closed form of LDHA. The minimal activities of such inhibitors againstsuggest that mobile loop closure may amplify differences between rather conserved active sites in human LDHA andLDH, pointing to the open conformation for development of effective chemotherapeutics against. These differences are also reflected in the observation that the two enzymes display distinct preferences for the two enantiomers of gossypol. While human LDHA is more sensitive to its levorotatory form,LDH apparently is inhibited more effectively by dextrorotatory gossypol. These observations will guide modeling studies to develop more effective anti-chemotherapeutics.
B. burgdorferi ’s reduced genome leaves only LDH as a means of utilizing pyruvate, making off-target effects significantly less likely [ B. burgdorferi growth. A potential limitation of these conclusions is the possibility that growth reduction was not caused by LDH inhibition, but was instead the result of off-target effects. This is a valid concern, somewhat mitigated by the fact that several mechanistically distinct LDH inhibitors (gossypol, oxamate, and galloflavin) displayed dose-dependent (albeit variable) growth suppression effects. Nonetheless, currently available LDH inhibitors have notable off-target effects. Oxamate, for example, is a pyruvate analog [ 38 ]. Due to this, oxamate can inhibit aspartate aminotransferase, impact the function of malate dehydrogenase, and otherwise disrupt cellular processes requiring pyruvate [ 39 ]. The effects might be concerning if focusing on other organisms, but’s reduced genome leaves only LDH as a means of utilizing pyruvate, making off-target effects significantly less likely [ 7 ]. As such, the presence of dose-dependent growth responses to oxamate addition suggests that LDH inhibition alone is sufficient to reducegrowth.
B. burgdorferi has a repertoire of genes to protect itself from common reactive oxygen species but remains susceptible to some forms of oxidative damage [ Borrelia turicatae . A distant relative within the Borrelia genus, B. turicatae has recently been found to be substantially more resistant to oxidative damage than B. burgdorferi [ Borrelia species have a similarly reduced genome to B. burgdorferi , lacking many metabolic pathways and likely relying on the same mechanisms for energy production [ B. turicatae culture reduces bacterial growth to a comparable extent, it may indicate that the reduction in B. burgdorferi growth is at least partly due to LDH inhibition. Concerns regarding off-target effects of gossypol are less easily dismissed. In addition to LDH inhibition, gossypol is known to cause oxidative damage to cells [ 40 41 ].has a repertoire of genes to protect itself from common reactive oxygen species but remains susceptible to some forms of oxidative damage [ 42 ]. One possible way to test whether gossypol inhibits growth through oxidative damage, LDH inhibition, or some combination thereof could be to evaluate the effects of gossypol on. A distant relative within thegenus,has recently been found to be substantially more resistant to oxidative damage than 43 ]. Overall,species have a similarly reduced genome to, lacking many metabolic pathways and likely relying on the same mechanisms for energy production [ 44 ]. If the addition of gossypol toculture reduces bacterial growth to a comparable extent, it may indicate that the reduction ingrowth is at least partly due to LDH inhibition.