A tiny brain implant, no bigger than a blueberry, is moving from lab concept to human testing in the United States. Motif Neurotech has secured clearance from the U.S. Food and Drug Administration to begin its first clinical trial targeting treatment-resistant depression. The decision marks a key step for a company aiming to bring engineering-driven solutions into mainstream mental health care. Nearly 3 million Americans live with forms of depression that do not respond to standard treatments. Motif’s approach focuses on directly interacting with neural circuits rather than relying only on drugs or talk therapy. The trial will test whether targeted electrical stimulation can improve outcomes where other methods have failed.
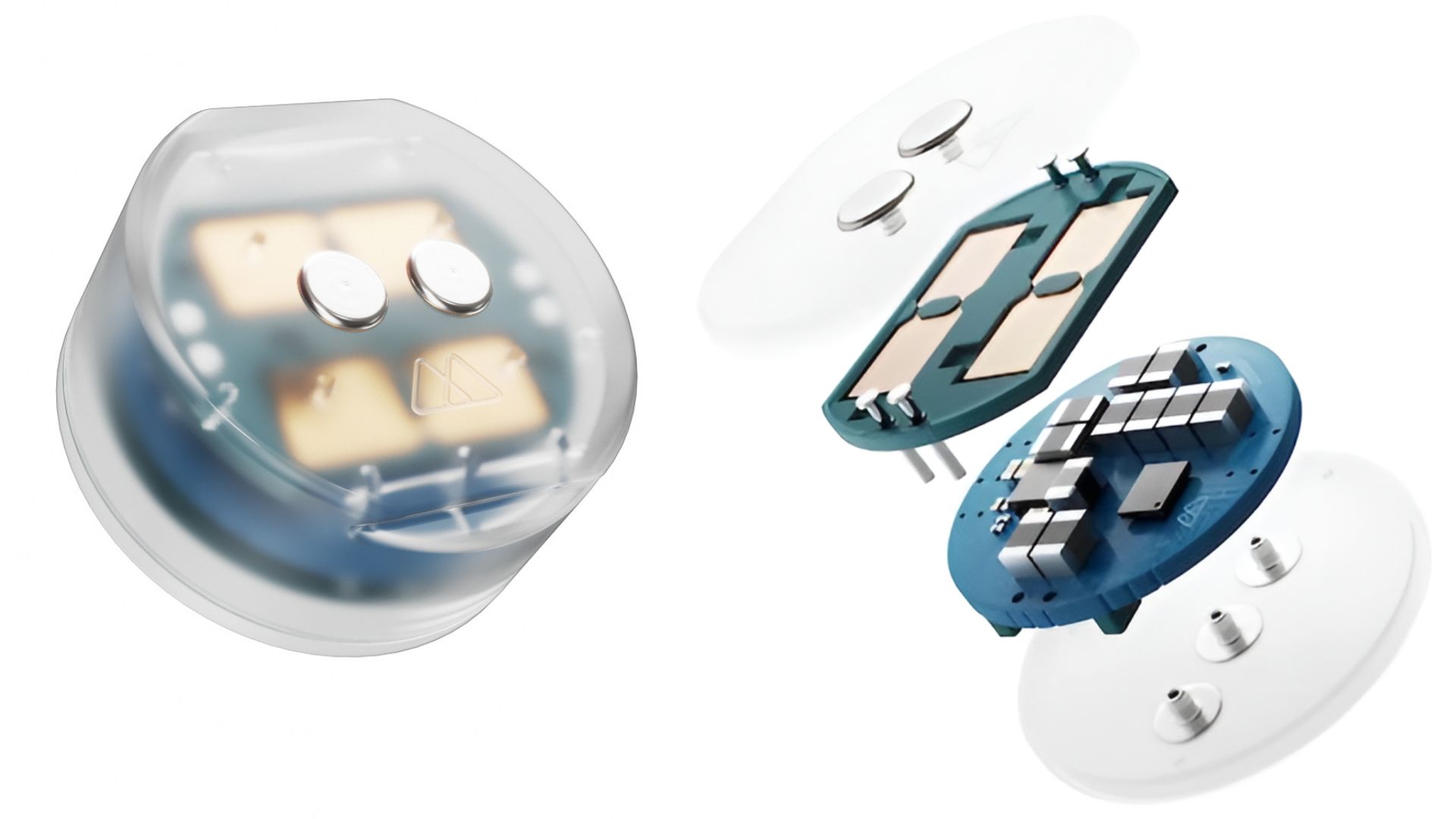
Minimally invasive brain device The company’s device, called Digitally programmable Over-brain Therapeutic (DOT), delivers electrical stimulation to brain circuits linked to depression. Unlike traditional implants, DOT does not penetrate brain tissue. Surgeons place it in the skull above the dura, reducing the risks tied to invasive neurosurgery. Motif engineered the system to run wirelessly, eliminating the need for implanted batteries or wired connections. The compact design aims to simplify procedures and improve patient comfort. Engineers also built the system to allow precise, programmable stimulation over time. “The goal for this technology is that it would be the mental health equivalent of a continuous glucose monitor for diabetes,” said Jacob Robinson, professor at Rice University and co-founder and CEO of Motif. “What has been really special for me personally on this journey is to be able to work all the way from a concept through the process of research and development funded by the federal government at Rice, and take that into a product that is going to affect people’s lives for the better.”
Clinical trial across US centers The early feasibility study will enroll adults whose depression has not improved after trying multiple therapies. Researchers will evaluate safety, device performance, and early signs of effectiveness. The study represents the first real-world test of the DOT system in patients. Several leading medical institutions will take part in the trial, including Baylor College of Medicine and Massachusetts General Brigham, along with other major centers across the country. These collaborations aim to ensure rigorous data collection and diverse patient representation. Motif obtained its investigational device exemption in just four years after founding. That pace stands out in the brain-computer interface field, where development timelines often stretch much longer due to regulatory and technical hurdles. Federal push for precision care Motif also joined a federal initiative focused on advancing behavioral health treatments through better data. The program, led by the Advanced Research Projects Agency for Health, supports faster validation of emerging therapies.
Through this effort, Motif will gather additional patient data alongside its clinical trial. Researchers aim to identify which individuals respond best to neuromodulation and how treatment effects evolve over time. “The idea with this funding is to support a number of teams who have rapid-acting interventions for a mental health condition and to collect additional data to help determine with greater precision whether a treatment is working, how it is working and which patients are benefitting most from which course of treatment,” Robinson said. The technology builds on more than a decade of research from teams at Rice and collaborating institutions. Federal agencies, including the National Institutes of Health and the Defense Advanced Research Projects Agency, supported early work. The project reflects a broader push to translate advances in neuroscience and engineering into practical therapies.
If you or someone you know is struggling with depression, support is available, and you don’t have to face it alone. U.S.: Call or text 988 (Suicide and Crisis Lifeline).
UK: Call 116 123 (Samaritans).