Despite the few studies that exist on the antiviral activity of cannabinoids, some research has been conducted that demonstrates their high anti-inflammatory and immunosuppressive power [ 2 10 ]. Thus, by inhibiting both the coronavirus replication processes and the inflammation provoked, there is the possibility of developing an effective therapeutic strategy [ 6 7 ]. The aim of this article is to review the therapeutic applications of cannabis derivatives and their applications for the treatment of COVID-19, based on the mechanisms through which they exert their pharmacological activity.
To date, seven strains of human-infecting coronaviruses have been identified [ 8 ]. The currently available treatment is supportive, as vaccination prevents the severe illness and death of patients, but its efficacy in controlling virus spread and preventing future deaths is not clear yet [ 9 ]. For these reasons, it has become a global public health emergency, making it extremely important to discover new alternatives for effective treatment.
The use of cannabis products has dramatically increased in recent times, due to the lack of response to standard treatments, for example to anxiety, chronical inflammation problems and seizures, which puts patients’ lives at risk. The hemp plant has been used for centuries in medicinal and recreational products such as hashish or marijuana. Cannabis use for medicinal purposes is legal in some countries, including Germany, Austria, Canada, Spain, Finland, Israel, Italy, Holland, Portugal and some states of the United States of America [ 1 ]. In Latin American countries, its use is permitted in Uruguay, Colombia and Argentina [ 2 ]. In Chile, the sale of cannabis-based medicines was authorized after the modification of a restricting law in 2015. For these reasons, it is expected that by 2025 the global medical cannabis market will reach43 billion [ 3 ]. The positive effects of the medicinal use of marijuana in conditions such as chronic pain, multiple sclerosis, nausea, vomiting, terminal diseases and epilepsy have been reported [ 1 2 ]. Several studies suggest that cannabidiol (CBD) may be useful in the treatment of patients with COVID-19 by inhibiting the expression of the protein critical for SARS-CoV2 virus replication and by binding as an agonist to the cannabinoid receptor 2 (CB-2), thus reducing inflammation and lung damage [ 4 7 ].
Alternatively to the aforementioned hydrolysis routes, AEA and 2AG can be oxidized by cyclooxygenase-2 (COX-2), different lipoxygenases or cytochrome P450 [ 23 ]. 2-AG hydrolysis inhibition could be an anti-inflammatory strategy to increase 2-AG levels and decrease eicosanoids levels. Human leukocytes use AA and unsaturated fatty acids to biosynthesize 2-AG and other monoacylglycerols (MAGs), which might decrease inflammation processes [ 24 ].
2-Arachidonoyl glycerol (2-AG) was the second endocannabinoid to be discovered. It is the most abundant endogenous cannabinoid in the CNS. It is an ester derived from arachidonic acid and glycerol, produced by the enzyme diacylglycerol lipase alpha and beta (DAGL). The inhibition of the actions of this endocannabinoid is mediated by hydrolysis with the enzyme monoacylglycerol lipase (MAGL) producing arachidonic acid (AA) and glycerol ( Figure 3 ). 2-AG behaves as a full agonist of the CB1 and CB2 receptors. However, it also binds to other receptors with cannabimimetic cellular activity, such as GRP55 and TRPV1 receptors [ 16 22 ].
In patients with autism spectrum disorder, the plasma concentrations of AEA are significantly lower compared to control children, suggesting that AEA signaling participates in the pathophysiology of autism, evaluated in 116 children around 3 to 12 years old [ 17 ]. The pharmacological increase in AEA levels results from the inhibition of fatty acid amide hydrolase (FAAH), which is responsible for AEA degradation to arachidonic acid (AA) and ethanolamine. Elevated levels of AEA may aid the treatment of post-traumatic stress disorder [ 18 ]. For example, in a double-blind, placebo-controlled study, the inhibition of FAAH (PF-04457845, 4 mg/once daily, over 10 days,= 16) increased 10-fold in baseline anandamide concentration, with an improvement of fear extinction memories in the patients and attenuation of the anxiogenic effects of stress [ 19 ]. Further, increased AEA concentrations caused by the inhibition of FAAH could promote learning during psychotherapeutic interventions [ 20 ]. On the other hand, AEA production appears to be deficient in patients with hypertrophic scars suggesting a correlation between the systemic and local skin endocannabinoid systems during human wound healing [ 21 ].
Anandamide (AEA) was the first discovered endocannabinoid from pig brain in 1992 [ 15 ]. Anandamide is produced from-arachidonoyl phosphatidylethanolamine, as shown in Figure 2 . It behaves as a full or partial agonist of the cannabinoid receptor 1 (CB1) as well as a partial CB2 agonist. It is present in the CNS, spleen, heart, testis, uterus and vascular endothelium of humans [ 16 ].
These natural molecules are derived from membrane phospholipids, which include arachidonic acid as endogenous cannabinoids present in the organism of all animals. They are synthesized in neurons on demand and released into the synaptic space, where they activate membrane receptors, acting in the vicinity of the site where they are released, regulating synaptic transmission. The principal endocannabinoids are anandamide (AEA) and 2-arachidonoyl glycerol (2-AG), but they also include 2-arachidonoyl-glycerol (2-AGE), O-arachidonoyl-ethanolamine (virodamine) and N-arachidonoyl-dopamine (NADA) [ 11 ]. The structures of endocannabinoids are given in Figure 1
The endocannabinoid system is a neuromodulatory network involved in many cognitive and physiological processes, such as in anti-convulsant, anti-inflammatory, anti-depressant and anti-tumorigenic actions, as well as exerting analgesic, hyperphagic, hypophagic, neuroprotective and antiemetic activities [ 11 ] and appetite regulation [ 12 ]. The endocannabinoid system is composed of endogenous cannabinoids (chemical derivatives of arachidonic acid), cannabinoid receptors and enzymes that produce and degrade endocannabinoids [ 13 14 ].
The GRP55 receptor is a putative cannabinoid receptor 3 (CB3). Experimental evidence links the GPR55 receptor to pathological conditions, providing a therapeutic target in cancer, neurodegenerative and metabolic disorders as well as in the regulation of microglia-mediated neuroinflammation [ 31 ]. The receptor has been found in the adrenal glands, spleen, intestine and CNS. The expression of the active receptor has been detected in bone cells [ 32 33 ]. GRP55 receptor activation occurs through phospholipase C (PLC)-β pathways. Consequently, there is an increase in cytosolic calcium concentration with the further stimulation of GTPases (Rac1, RhoA and Cdc42) and mitogen- or stress-activated protein kinases (MAPKs, ERK and p38) and NFAT, NFKB, CREB and ATF2 transcription factor activation [ 34 36 ]. GRP119 cannabinoid receptors are localized in humans predominantly in the pancreatic tissue and in the enteroendocrine L cells of the gastrointestinal tract. Their activation elevates plasma insulin concentrations in mice and decreases food intake and weight in rats. It has been proposed as the endogenous receptor for oleoyl ethanolamide [ 37 ].
CB2 receptors are expressed by immune cells, such as leukocytes, in the spleen, tonsils and in the peripheral nervous system. The functions of CB2 receptors include the modulation of the immune system and inflammation by the release of cytokines. Since selective CB2 receptor agonists do not cause psychotropic effects, these are increasingly investigated for the therapeutic applications of cannabinoids, such as analgesics, anti-inflammatories and anti-neoplastics [ 29 30 ].
CB1 receptors are principally expressed in the neurons of the cortex, spinal cord and peripheral nervous system as well as in peripheral organs and tissues. These receptors are expressed in brain areas responsible for memory processing, movement, and pain modulation. Their activation results in different effects on pain sensation as well as on sleep, memory, appetite, mood, and motor regulation [ 29 30 ].
In 1990, the first cannabinoid-specific receptor (CB1) was identified and cloned, followed by a second receptor (CB2). Cannabinoid receptor 1 and CB2 differ in their signal transduction and in their distribution in different tissues. The identification of these receptors led to a better understanding of phytocannabinoid and endocannabinoid functions and to the creation of synthetic cannabinoids [ 25 ]. Cannabinoid binding to their receptors results in distinct effects depending on which signaling pathway is activated. Signaling pathway activation depends on the agonist structure and the monomeric or heteromeric cannabinoid receptor states [ 26 ]. Furthermore, CB1 and CB2 receptors differentially regulate TNF-α-induced apoptosis in HT22 hippocampal cells, CB1 being a promoter of signaling and neuroprotection [ 27 ]. Once endocannabinoids have been released by the presynaptic neuron, the cannabinoid receptors are activated, inhibiting the release of neurotransmitters, such as glutamate [ 28 ]. This action of cannabinoids confers to them important physiological roles as regulators of synaptic activity.
Δ8-THC and CBD are found in some plant varieties and always in low concentrations. CBD also exerts psychoactive properties, and its action on the CB2 receptor in splenocytes and thymocytes causes a decrease in the transcription of the gene for interleukin-2 (IL-2) [ 43 ]. CBD has shown anxiolytic, anticonvulsant and antipsychotic effects. Further antioxidant, anti-inflammatory, antiemetic, antitumor and neuroprotective properties of CBD have been reported in preclinical studies. Cannabinol has analgesic properties with fewer psychoactive effects than Δ9-TCH. The activity of commercial formulations based on cannabinoids is summarized in Table 1
Despite THC displaying the highest psychoactive potential, its pharmacological properties have been proven in clinical and preclinical assays as appetite stimulation, the suppression of chemotherapy-induced nausea and vomiting and the inhibition of pain, spasticity, neuroprotective, anti-inflammatory and antitumor effects [ 40 42 ].
Cannabis plants contain over 500 bioactive compounds in their 18 different species, including over 100 different naturally occurring phytocannabinoids. Phytocannabinoids are produced in glandular hairs or trichomes. While they are found on most aerial surfaces of the plant, phytocannabinoids are primarily located in the bracts and flowers of the plant. [ 30 ] Phytocannabinoids are compounds of terpene phenolic structure with 21 carbon atoms. The main phytocannabinoids are Δ9-tetrahydrocannabinol (THC), Δ8-tetrahydrocannabinol (Δ8-THC), cannabidiol (CBD), cannabinol (CBN), and CBD being the known active compounds of pharmacological plant activity ( Figure 4 ).
The genuscomprises the plantsandgrows in tropical and subtropical regions as well as temperate climate zones, e.g., in Europe [ 1 2 ]. The original plant has been known for 10,000 years in practically all cultures since the discovery of agriculture. Countries such as China, India, Turkestan and Arabia have used its compounds for medicinal purposes to treat diseases such as malaria, beriberi, constipation, rheumatic pains, headaches, female ailments and other conditions [ 11 ]. In the 19th century, its widespread use as a stimulant, sedative and analgesic became popular [ 30 ]. In 1860, the first committee of physicians in the USA was created to systematically study its use and properties. In 1964, delta-9 tetrahydrocannabinol (Δ9-THC), the main substance responsible for the psychoactive and pharmacological properties of marijuana extracts, was isolated.
Another issue of great importance is the recently discovered interaction between first generation antiepileptic drugs (carbamazepine, oxcarbazepine, phenytoin, phenobarbital and primidone) and the decrease in the concentration of some drugs used for COVID-19 treatment (atazanavir and remdesivir, darunavir/cobicistat and lopinavir/ritonavir). Therefore, in these cases, the use of CBD to treat both conditions can be an alternative that can change the results obtained [ 64 ].
Cannabinoids are mostly used as anxiolytic, relaxing and anti-inflammatory natural agents with adjuvant properties for the treatment of epilepsy, schizophrenia, multiple sclerosis, depression or chronic pain [ 38 55 ]. In the case of epilepsy, it has been shown that the brain tissue of these patients shows an overexpression of proinflammatory cytokine IL-1β and IL-6 genes together with nuclear transcription factor kappa B (NFKB) [ 56 ]. Cannabinol can inhibit the G protein-coupled orphan receptor (GRP55) and decrease NFKB signaling, the latter probably by binding to nuclear PPAR-g receptors, thus reducing the expression of proinflammatory enzymes such as iNOS (nitric oxide synthase) and COX-2 (cyclooxygenase type 2) and metalloprotease and proinflammatory cytokine production [ 10 58 ].
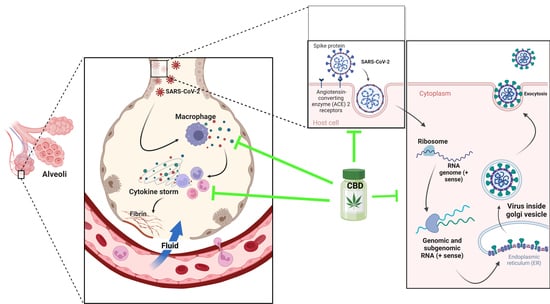
The angiotensin-converting enzyme 2 (ACE 2) has been pointed out as the principal receptor for the interaction of SARS-CoV 2 with human cells, which is mainly expressed in type I and II pneumocytes, making these cells are more susceptible of being infected by the virus. ACE 2 receptors are also expressed by lung AT2, liver cholangiocytes, colon colonocytes, esophageal keratinocytes, ileum EC, rectum EC, stomach epithelial cells and kidney proximal tubules [ 70 ]. CD169macrophages residing in the spleen and lymph node tissues can be infected [ 71 75 ]. Alveolar macrophages do not only protect the lungs from pathogens, but also participate in enhancing specific T-cell responses, repairing damaged tissue and recruiting neutrophils leading to increased inflammatory cell traction [ 76 78 ]. The hyperactivation of lung macrophages and proinflammatory monocyte-derived macrophages (MDM) occurs in the small airways. The transcriptomic analysis of plasma and bronchoalveolar lavage fluids from SARS-CoV2 patients has demonstrated large amounts of proinflammatory cytokines, chemokines and soluble inflammatory mediators, including TNF-α, IL-6, IL-1β, IL-2R, IL-8, inducible protein (IP)-10, C-reactive protein and D-dimer, culminating in a cytokine storm [ 79 ]. In the inflammation process, the overexpression of genes that express chemokines was observed, which are critical for recruiting neutrophils (CXCL17) and monocytes (CCL2, CCL7) in the lungs [ 80 ]. In SARS-CoV2 and MERS-CoV, a rapid and specific memory CD8 T-cell response is required to protect against infection [ 24 25 ]. However, severe SARS-CoV-2 infection results in CD4 and CD8 T-cell lymphopenia and a decrease in INF-γ-producing T cells [ 81 82 ].
The seriousness of SARS-CoV2-caused illness lies in its capability of inducing in the infected person a rapid and intense immune response that courses in an uncontrolled manner, unleashing a storm of cytokines resulting in a variety of symptoms, including severe morbidity and mortality. This cytokine storm is produced by the organism attempting to defend itself against viral invasion, thus stimulating the production of macrophages, interleukins, chemokines and proinflammatory mediators [ 66 ]. The endocannabinoid system has the role of acting as a key regulator of the immune system, as it possesses the capacity of immunosuppression and repressing cytokine cascade, inhibiting immune cell proliferation and antibody production [ 7 70 ].
As of 24 October 2022, there have been 624,235,272 confirmed cases of COVID-19, including 6,555,270 deaths, reported to the WHO, and a total of 12,814,704,622 doses of vaccine have been applied [ 65 ].
7. Indispensable Proteins for the Development of Infection and CBD
85, The structural characteristics of SARS-CoV2 indicate that there are host cell proteins that are indispensable for the progress of viral infection. Once inside the lung cells, it binds to the host cell membrane receptor: angiotensin-converting enzyme 2 (ACE 2), via the transmembrane spike glycoprotein (structural glycoprotein of the virus) and induces its entry through membrane endocytosis. This glycoprotein forms homotrimers that protrude from the surface of SARS-CoV2. The subunits of the SARS-CoV2 spike glycoprotein trimer consist of an S1 subunit that binds to host cell ACE2 to initiate infection, an S2 subunit that mediates the fusion of the virus with host cells and a transmembrane domain [ 83 ]. While ACE2 is the receptor for viral entry, the transmembrane enzyme transmembrane serine protease 2 (TMPRSS2) primes viral spike proteins for SARS-CoV2 entry into host cells. Its function is given by proteolysis and the consequent activation of the spike protein by cleaving it into two covalently linked peptides: the S1 (ACE2 receptor-binding terminal) and S2 (viral cell membrane fusion-mediated terminal) sites or subunits, leading to conformational changes for the fusion of the virus with the host membrane and the virus entering of the cytoplasm [ 84 86 ]. The S1 subunit is primarily responsible for host virus range determination and cell tropism [ 71 87 ].
88, Then, ORF1, one of the nonstructural viral proteins, stimulates virus replication and RNA synthesis, while the nucleocapsid phosphoprotein (structural viral protein) packages the viral genome, forming a helical nucleocapsid with an RNA chaperone function. The surface glycoproteins evoke virion assembly and morphogenesis, producing virus particles and releasing them by exocytosis [ 66 89 ]. Any interference with the expression of one of these proteins may disrupt the transmission cycle of SARS-CoV2 [ 6 ].
C. sativa compounds have strong binding affinities to the initiation and termination codons of ORF1, the surface glycoprotein, the envelope protein and nucleocapsid phosphoprotein mRNAs of the complete SARS-CoV2 genome isolated from KwaZulu-Natal, South Africa [ Erukainure et al. demonstrated thatcompounds have strong binding affinities to the initiation and termination codons of ORF1, the surface glycoprotein, the envelope protein and nucleocapsid phosphoprotein mRNAs of the complete SARS-CoV2 genome isolated from KwaZulu-Natal, South Africa [ 90 ]. This highlights the utility of phytocannabinoids in slowing down virus replication, translation, assembly, and release. The nature of these binding affinities could be due to their chemical properties. The cannabinoid compounds present in their structure free hydroxyl group and aromatic rings that enable them to interact with purines, pyrimidines or the phosphate terminus of the mRNA sequence of the SARS-CoV2 genome through hydrogen bonds or intermolecular interaction.
50 of 8 µM. In view of that, CBD is at least as potent as the antiviral drugs Remdesivir (RDV) and Lopinavir, which are employed for COVID-19 treatment [95,96, Purines and pyrimidines, as part of nucleotides, are capable of undergoing nucleophilic or electrophilic reactions, specifically the formation of hydrogen bonds between the oxygen at carbon 2 of the purines in the SARS-CoV2 mRNA genome [ 91 ]. The series of electrostatic interactions between the pyrimidines and the natural product could be due to the free amine bound to carbons 2 and 6 of guanine and adenine, respectively. Similarly, the polar region of phytocannabinoids could electrostatically interact with the phosphate terminal of SARS-CoV2 mRNA [ 92 93 ]. CBD inhibits SARS-CoV2 replication with an ICof 8 µM. In view of that, CBD is at least as potent as the antiviral drugs Remdesivir (RDV) and Lopinavir, which are employed for COVID-19 treatment [ 94 97 ].
CBD effectively eradicates viral mRNA expression in host cells. SARS-CoV2-induced gene expression reduction connected to chromatin modification and transcription was reversed. Gene expression reduction of mRNA spike, membrane, envelope and nucleocapsid protein-coding mRNAs was observed following 24 h of CBD treatment. It also suggested that CBD acts to prevent viral protein translation and the associated cellular changes [ 98 ]. As underlying mechanisms, CBD may suppress viral infection and promote viral RNA degradation through the induction of the interferon signaling pathway. CBD may also reduce the viral titer to allow the normal host activation of the interferon pathway, which is suppressed upon infection by SARS-CoV2 [ 99 100 ].
®) show a maximum concentration (C max ) of CBD in blood plasma in the nM range, while 7-OH-CBD has a Cmax at μM concentrations. These results suggest that CBD itself is not present at sufficient concentrations for the effective inhibition of the SARS-CoV2 transmission cycle in humans. In contrast, the plasma concentrations of its metabolite 7-OH-CBD, whose C max is augmented following co-administration of CBD with a high-fat meal, are sufficient for the potential blockade of human SARS-CoV2 infection [ CBD is metabolized to 7-carboxy-cannabidiol (7-COOH-CBD) and 7-hydroxy-cannabidiol (7-OH-CBD) in the liver and intestine. CBD and 7-OH-CBD inhibit SARS-CoV2 replication in A549-ACE2 cells (EC50 of 3.6 μM) [ 98 ]. The levels of these cannabinoids in healthy patients taking CBD (Epidiolex) show a maximum concentration (C) of CBD in blood plasma in the nM range, while 7-OH-CBD has a Cmax at μM concentrations. These results suggest that CBD itself is not present at sufficient concentrations for the effective inhibition of the SARS-CoV2 transmission cycle in humans. In contrast, the plasma concentrations of its metabolite 7-OH-CBD, whose Cis augmented following co-administration of CBD with a high-fat meal, are sufficient for the potential blockade of human SARS-CoV2 infection [ 101 ].
The crystal structure of the SARS-CoV2 spike protein (C-terminal domain) in complex with the ACE2 receptor (human) was solved in 2020, giving the opportunity to understand the interaction between the virus and the human receptor [ 102 ]. This knowledge gives opportunities to create selective ligands targeting the viral S1 protein and preventing human cells from SARS-CoV2 infection [ 5 ]. In this sense, cannabidiolic acid (CBDA) and cannabigerolic acid (CBGA) showed high binding affinity to recombinant S1 [ 103 ].
50 of 7.91 µM [106, On the other hand, in silico analysis suggests that CBD binds to the S1 virus membrane protein by interactions with the residues Q189, M165 and E16, causing an inhibitory effect with an ICof 7.91 µM [ 104 ]. On the other hand, ACE2 expression in the epithelium is high in males, older people and smokers, evidencing the risk of the population to the expression of the ACE2 receptor [ 105 ]. Downregulation of ACE2 levels due to CBD actions could be a strategy to decrease susceptibility for virus infection. CBD decreases the expression of ACE2 and TMPRSS2 in oral, lung and intestinal epithelia, which could reduce the burden of the virus [ 71 107 ]. A study conducted in different tissues (EpiAirway TM, EpiAirwayFT TM, EpiOral TM, EpiIntestinal TM) with a level of ACE2 and TMPRSS2 overexpression identified the significant effects of cannabis in decreasing ACE2 and TMPRSS2 expression levels [ 107 ]. This information suggests that phytocannabinoids may prevent SARS-CoV2 infection.
To determine the mechanisms by which the expression downregulation of ACE2 and TMPRSS2 takes place, the mRNA levels of the genes miR-200c-3p and let-7a-5p encoding ACE2 and TMPRSS2 were measured. qRT-PCR showed that CBD modulated ACE2 and TMPRSS2 expression levels through transcriptional and post-transcriptional mechanisms. A possible mechanism by which they obtain these effects is through the activation of the AKT pathway participating in the post-transcriptional regulation of ACE2 and TMPRSS2 expression. CBD significatively decreased p65 (p-p65, Ser536) and phosphorylated AKT1/2/3 (p-AKT1/2/3) levels [ 108 ].