Cannabis is an annual plant that can be grown efficiently indoors under controlled conditions or outdoors under full spectrum sunlight [ 11 ]. Secondary metabolism in cannabis plants is influenced by several environmental cultivation conditions. To date, the effects of outdoor cultivation factors compared to indoor conditions on the cannabinoid and terpene profiles in cannabis have not been fully studied.
During inflorescence, the cannabis plant produces a plethora of cannabinoids and terpenes in the glandular trichome cells [ 26 ]. It is staggering and remarkable the number of these molecules that the plant produces. There is also added complexity that the cannabinoids can be oxidized in a multitude of ways. This creates two types of cannabinoids. The first are ones that are intrinsic to the cannabis plant because they are made by a biological pathway in the plant [ 27 ]. We refer to these as intrinsic cannabinoids. There are also other cannabinoids that are extrinsic to the cannabis plant that are created through subsequent reactions due to their environment, such as oxidation or photochemistry. We refer to these as extrinsic cannabinoids. The terpenes broadly fall into four main categories: monoterpenes (10 carbon), monoterpenoids (oxygenated terpenes), sesquiterpenes (15 carbon), and sesquiterpenoids (oxygenated sesquiterpenes) [ 27 ].
In this study, we used commercial cannabis samples that are cloned from a common parent but which are grown both indoor and outdoor under optimized conditions. The outdoor samples were grown in raised beds using a proprietary mixture of all-natural soil and composts under full sunlight. The indoor samples were grown under artificial light in a proprietary growth medium. The outdoor samples were stickier to the touch and were much more pungent than the indoor samples. The morphology and color of the flowers were similar. Each of the samples was from the same season to eliminate issues of large differences in age between the samples. Therefore, we can assess the importance of the two environments on the terpenes and cannabinoids metabolite compositions in two cultivars.
Although being genetically identical, the indoor and outdoor samples from either of these two cultivars are completely distinguished by the composition of their cannabinoids. While we are applying this analysis here to different methods of cultivation, this type of analysis of the cannabinoids will enhance our understanding of the effects the environment and genetics have on the cannabinoids produced and also in the understanding of how to classify the multitude of different cultivars of cannabis that are commercially available.
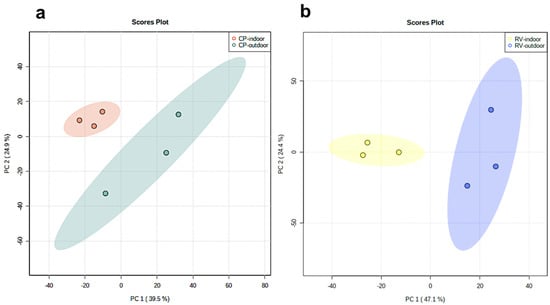
We performed an untargeted LC-MS-based metabolomic to analyze the metabolic profile of two cultivars, CP and RV, each genetically identical produced through clones, grown either indoors or outdoors under optimized conditions ( Supplementary Figure S1a ). We analyzed three samples of each (n = 3) for both indoor and outdoor samples. The untargeted LC-MS analysis of the samples resulted in the detection of 1001 and 1316 features in the positive and negative ESI modes, respectively. Unsupervised principal component analysis (PCA) of the extracted features showed tight clustering of QC samples and clear serrations of indoor versus outdoor groups ( Figure 1 and Supplementary Figure S1b ). The tight clustering of the multiple injections of the QC sample implies no drift over time and confirms the reproducibility of the LC-MS system. We further examined the data to ensure that no terpenes or flavonoids were present in this analysis. The PCA score plots in the negative mode indicate the differences in the cannabinoid profiles ( Figure 1 ). The PC1 (describing the variation between groups) from the two plots was found to be 39.5% and 47.1%, and PC2 (which describes the variation within the groups) was 24.9% and 24.4% for the CP and RV models, respectively. Thus, the PCA score plots represents a distinctive clustering due to metabolic differences. Supplementary Figure S2a,c show the loadings plots, displaying the discriminative ion features localized in the peripheral (extreme values in PC1 and PC2) areas of the plots between indoor-grown and outdoor grown cultivars.
2.2. Cannabinoid Analysis
9-THCA, and CBDA for CP ( To further understand the differences between the molecular profiles that gave rise to the disparate PCA results when comparing indoor versus outdoor cultivation, we conducted targeted cannabinoid analysis of the primary, intrinsic cannabinoids, CBGA, CBCA, Δ-THCA, and CBDA for CP ( Figure 2 a) and RV ( Figure 2 b).
9-THCA, which are enhanced and depleted significantly in the RV-outdoor samples, respectively. This series is of importance because CBGA is a common precursor to CBCA, Δ9-THCA, and CBDA that occur via three different biochemical pathways by particular synthases, among which the most prominent are tetrahydrocannabinolic acid-, cannabidiolic acid-, and cannabichromenic acid-synthase, leading to the production of THCA, CBDA, and CBCA, respectively [ We found little difference between the indoor- and outdoor-grown samples for these primary cannabinoids except CBCA and Δ-THCA, which are enhanced and depleted significantly in the RV-outdoor samples, respectively. This series is of importance because CBGA is a common precursor to CBCA, Δ-THCA, and CBDA that occur via three different biochemical pathways by particular synthases, among which the most prominent are tetrahydrocannabinolic acid-, cannabidiolic acid-, and cannabichromenic acid-synthase, leading to the production of THCA, CBDA, and CBCA, respectively [ 28 29 ]. IN addition, then they can be decarboxylated through various processes such as light exposure, heating, or through chemical reactions [ 30 ].
9-THC was lower while the level of CBG was considerably higher in CP-outdoor samples. Δ9-THC is the primary psychotropic metabolite of cannabis and binds to specific G-protein-coupled receptors, cannabinoid CB1 and CB2 receptors [38,39, The corresponding data for the decarboxylation products of these primary cannabinoids is shown in the Supplementary Figure S3 and follows the same trends seen in Figure 2 , albeit in much lower amounts. The level of Δ-THC was lower while the level of CBG was considerably higher in CP-outdoor samples. Δ-THC is the primary psychotropic metabolite of cannabis and binds to specific G-protein-coupled receptors, cannabinoid CB1 and CB2 receptors [ 31 ]. Although there is growing research on the potential value of THC in the treatment of a number of human diseases [ 26 32 ], its development as a therapeutic has been limited due to its psychoactive properties. However, CBD, CBG, and CBC have very low affinity for CB1/CB2 receptors and have less psychotropic activities compared to THC. We detected a significantly higher level of CBD in the RV-outdoor samples compared to the indoor-grown RV samples. CBD is one of the most abundant cannabinoids and is well-known for its anxiolytic and antipsychotic properties [ 33 ]. It has been shown that CBD has multiple pharmacological benefits in in vitro and animal studies, which makes it a very promising therapeutic commodity in inflammation, diabetes, cancer, and neurodegenerative diseases [ 34 35 ]. It is demonstrated that CBG has a promising therapeutic potential in the treatment of inflammatory bowel disease and prostate cancer [ 35 36 ]. An early study by Mahlberg and Hemphil showed the higher level of THC in cannabis leaves grown under sunlight than the plants grown under filtered green light and darkness while there was no significant difference in THC content in plants grown under filtered blue and red lights and shaded daylight compared to the sunlight grown plants [ 37 ]. Moreover, they showed that the level of CBC was maintained comparable or lower in plants grown under daylight than light stressed conditions. CBC is particularly abundant in young plants or freshly harvested dry-type cannabis and is hypothesized to be a synergist for the psychoactive cannabinoids [ 26 40 ].
p -value < 0.05 and fold change threshold of 2) in the indoor-grown CP and RV groups, respectively. Moreover, the relative abundance of 42 ion features was significantly lower in the indoor-grown CP samples ( A more detailed analysis of all the extracted signal intensities using volcano plots was performed to visualize independent changes in cannabinoid profile and to discriminate between outdoor and indoor-grown cultivars ( Supplementary Figure S2b ). The levels of 42 and 32 ion features were remarkably higher (FDR-corrected-value < 0.05 and fold change threshold of 2) in the indoor-grown CP and RV groups, respectively. Moreover, the relative abundance of 42 ion features was significantly lower in the indoor-grown CP samples ( Supplementary Figure S2c ). By removing different co-existing adduct ions, in-source fragment ions, and finally matching the MS/MS fragments with available commercial standards or reported in literature, we could annotate 21 unique cannabinoids as shown in Table S1 . Intriguingly, we found significant differences in the levels of the cannabinoids between the two cultivation methods that produced through the environment the cannabis is subject to during growth, curing, and packaging, as shown in Figure 3 . For both cultivars, the oxidation and degradation products of the primary cannabinoids, including CBN, CBNA, OH-CBNA, CBNBA, CBNDA, CBEA, CBT-isomer 1, and CBT-isomer 2, and others, are significantly amplified in the indoor-grown samples. CBNDA and CBEA are the results of full aromatization and photo-oxidation of CBDA, respectively. CBT isomers are the hydroxylated forms of THC [ 41 ]. CBN and its derivatives and analogs are synthesized from the oxidative aromatization of their corresponding THC-type derivatives [ 34 ]. The continued exposure of CBN to ultraviolet light in the presence of oxygen or air produces degradation products, OH-CBN. Interestingly, we found over two orders of magnitude more CBNA compared to CBN in these samples ( Figure 3 ), indicating that CBNA produced from THCA and consequently, CBNA becomes CBN through spontaneous or induced decarboxylation.
It should also be noted that CBNA is not currently being tested on the California Certificate of Analysis (COA) for cannabis. Therefore, consumers might be exposed to much more CBN after heating the samples than is depicted on the COA. Significant accumulation of CBNA and the plethora of other oxidation and degradation products such as OH-CBNA and CBNBA in the indoor samples is imperative because many of these oxidized cannabinoids might have diverse biological activities [ 42 ]. For example, it is shown that CBN is a strong sedative when it is combined with THC [ 43 ].