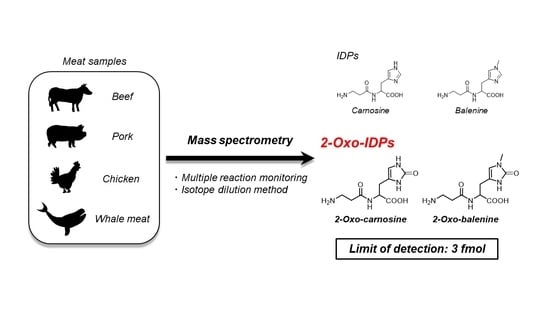
Numerous studies have demonstrated the detection and quantification of IDPs using HPLC coupled with various detection systems, such as ultraviolet (UV) detectors [ 26 27 ] and fluorescence detectors [ 28 29 ]. Furthermore, HPLC coupled with MS/MS has also been applied for the detection and quantitation of IDPs [ 30 ]. However, a highly sensitive detection system for 2-oxo-IDPs is lacking. In this study, we developed a sensitive, highly selective, and absolute quantitative analysis method for 2-oxo-IDPs using HPLC-ESI-MS/MS coupled with a stable isotope dilution method. We also synthesized stable isotope-labeled IDPs and 2-oxo-IDPs as standards. Furthermore, using stable isotope labeled-standards and the newly developed method, we quantified the IDP and 2-oxo-IDP contents in several meat samples, such as beef, pork, chicken, and whale meat.
Recently, we revealed that 2-oxo-imidazole-containing dipeptides (2-oxo-IDPs), such as 2-oxo-carnosine, 2-oxo-anserine, 2-oxo-homocarnosine, and 2-oxo-homoanserine, are endogenously produced in mouse tissues as a novel oxidized derivative of IDPs by high-performance liquid chromatography with online electrospray ionization-tandem mass spectrometry (HPLC-ESI-MS/MS) coupled with a stable isotope dilution method [ 7 23 ]. 2-Oxo-carnosine and 2-oxo-anserine have also been detected in several types of meat samples [ 24 ]. Notably, multiple in vitro assays have demonstrated that 2-oxo-carnosine and 2-oxo-anserine exhibit a greater antioxidant capacity than that of corresponding IDPs, suggesting that 2-oxo-IDPs play a key role in the antioxidant capacity of IDPs [ 25 ]. In addition, 2-oxo-homoanserine also have showed potent radical scavenging capacity than the precursor, homoanserine [ 23 ]. These findings emphasize the potentials of the bioactive substances for developing preventive and therapeutic strategies for oxidative stress-related diseases such as Alzheimer’s disease. However, further studies to unveil the physiological functions of 2-oxo-IDPs are currently difficult due to the limited commercially availability of several precursor IDPs (i.e., balenine, homocarnosine, and homoanserine). Thus, the development of a simple and efficient procedure to prepare standards of IDPs and its oxidized derivatives, 2-oxo-IDPs, with high yield and high purity, is required.
Imidazole-containing dipeptides (IDPs) are endogenous dipeptides composed of histidine or its methylated derivatives. To date, five types of IDPs have been reported. Carnosine (β-alanyl--histidine), a representative IDP, was first discovered in beef extract in the 1900s [ 1 ], and its methylated analogs, anserine (β-alanyl-3-methyl--histidine) and balenine (β-alanyl-1-methyl--histidine) were identified in goose and snake samples [ 2 3 ]. Homocarnosine (γ-aminobutyryl--histidine) and homoanserine (γ-aminobutyryl-3-methyl--histidine), in which β-alanine is replaced by γ-aminobutyric acid (GABA), were first isolated from bovine brain samples [ 4 5 ]. Several IDPs are endogenously produced in vertebrates; they are abundant in the skeletal muscle and brain and have been detected in other tissues, such as the heart, kidneys, and liver [ 6 7 ]. The metabolism of IDPs is strictly controlled by specific enzymes, such as carnosine synthase, carnosine-methyltransferase, and carnosinase [ 8 10 ]. Accumulating evidence has indicated that IDPs are widely distributed and precisely regulated by enzymes, suggesting that they act as important functional molecules in vivo.
IDPs and 2-oxo-IDPs were quantified using a Shimadzu LCMS 8060 triple quadrupole mass spectrometer (Shimadzu, Kyoto, Japan) coupled with the Nexera X2 UHPLC system (Shimadzu) and an Intrada Amino Acid column (3.0 × 100 mm; Imtakt). A discontinuous gradient of solvent A (acetonitrile containing 0.1% formic acid) and solvent B (100 mM ammonium formate) was used as follows: 50% B at 0 min; 50% B at 10 min; 99% B at 11 min, at a flow rate of 0.6 mL/min for the separation of IDPs, and 30% B at 0 min, 35% B at 7 min, 99% B at 7.1 min, at a flow rate of 0.6 mL/min for the separation of 2-oxo-IDPs. The mass spectrometer was operated in the positive mode under the following conditions: nebulizer gas: 3.0 L/min; heating gas: 10 L/min; drying gas: 10 L/min; interface temperature: 350 °C; desolvation line temperature: 250 °C; heat block temperature: 400 °C. LabSolutions LCMS Ver. 5.91 (Shimadzu Scientific, Inc., Columbia, MD, USA) was used for data collection and quantification. Regression equations for the calibration curves were obtained using GraphPad Prism (GraphPad, Inc., La Jolla, CA, USA). The limit of quantification (LOQ) and the limit of detection (LOD) were calculated as the lowest concentration on the calibration curve that was linear and had a signal-to-noise ratio of >10 or >3, respectively.
Meat extracts were prepared as described previously [ 24 ], with slight modifications. In brief, meat samples (1 g) were shredded with scissors and homogenized using a Heidolph homogenizer (Heidolph, Schwabach, Germany) in a 10-fold volume () of 80% acetonitrile in water containing 50 pmol stable isotope-labeled standards on ice. Next, 50 µL of the homogenate was collected for the analysis of IDP contents and protein quantification and was centrifuged at 12,000×for 20 min at 4 °C. The supernatants were diluted with 0.1% formic acid in water containing 5 pmol stable isotope-labeled standards and subjected to HPLC-ESI-MS/MS. For the analysis of 2-oxo-IDPs, the remaining homogenates were centrifuged at 12,000×for 20 min at 4 °C, and the supernatants were purified using a DOWEX cation exchange column. The eluate was then dried in vacuo, dissolved in 100 µL of 0.1% formic acid in water, and subjected to HPLC-ESI-MS/MS.
The DPPH radical scavenging assay was carried out as previously described [ 7 25 ], with slight modifications. In brief, in a 96-well plate, 100 μM DPPH was incubated in 12 mM sodium phosphate buffer (pH 7.4) in the presence or absence of 2-oxo-IDPs (final 20–40 μM) or IDPs (final 200–1000 μM) for 20 min at room temperature (25 °C). Absorbance at 517 nm was measured using an Infinite 200 PRO microplate reader (Tecan, Männedorf, Switzerland). Trolox solutions (final 5–30 μM) were used for defining the standard curve. Radical scavenging capacity was evaluated by the inhibition ratio (%), which was calculated by the following formula: Inhibition ratio (%) = (Ac − As)/Ac × 100, where Ac and As indicate the absorbances of the blank control (water) and sample, respectively [ 25 ]. The radical scavenging capacity was expressed as Trolox equivalent antioxidant capacity (TEAC), which was calculated using the Trolox standard curve.
The synthesized or commercially available IDPs were oxidized by the copper/ascorbate oxidation system as described previously [ 7 ], with slight modifications. The aqueous mixtures (200 mL) containing 50 mM IDPs and 500 mM ascorbate were adjusted to pH 5.0 with 1 M NH. After the addition of 500 mM CuSO(2 mL), the mixtures were incubated under oxygen aeration for 90 min at room temperature (25 °C). After the oxidation of IDPs, the reaction mixtures were adjusted to pH 2.0 with 4 M HCl, and then the mixtures were applied to the DOWEX cation exchange column (10 mL) pre-equilibrated with 0.1 M HCl. The flow-through fractions containing 2-oxo-IDPs were collected and applied to another DOWEX cation exchange column (100 mL) pre-equilibrated with 0.1 M HCl. The column was washed with three column volumes of 1 M HCl, and then 2-oxo-IDPs were eluted with three column volumes of 1 M NH. After in vacuo concentration for NHremoval, the samples were diluted with 10 volumes of 80% acetonitrile in water and subjected to silica gel column (Purif-Pack, SI-25, size: 60; Shoko Scientific, Yokohama, Japan). The cartridge was washed with three column volumes of 70% acetonitrile and 2-oxo-IDPs were eluted with three column volumes of 60% acetonitrile. After concentration, the samples were purified by HPLC using a Scherzo SS-C18 column (10.0 × 150 mm; Imtakt) and a discontinuous gradient of solvent A (water containing 0.1% formic acid) and solvent B (water containing 50% acetonitrile and 100 mM ammonium formate) (0% B at 0 min; 20% B at 0.1 min; 20% B at 7 min; 100% B at 7.1 min) at a flow rate of 3.0 mL/min. After the concentration of the eluate containing 2-oxo-IDPs, the samples were diluted with 80% acetonitrile in water and purified by HPLC using an US-Amino column (10.0 × 100 mm; Imtakt) and a discontinuous gradient of solvent A (acetonitrile) and solvent B (water) (0% B at 0 min; 15% B at 0.1 min; 15% B at 5 min; 50% B at 20 min) at a flow rate of 3.0 mL/min. The elution was monitored by absorbance at 250 nm, and the fraction containing 2-oxo-IDPs was concentrated in vacuo. The purity of synthesized 2-oxo-IDPs were analyzed by HPLC-PDA analysis ( Supplementary Figure S1 ). The chemical structures of the products were characterized by HPLC-ESI-MS/MS. The concentrations of synthesized 2-oxo-IDPs were calculated as described above. Experimental procedures described in this section are shown in Supplementary Figure S3
Balenine, homocarnosine, homoanserine, and stable isotope-labeled balenine were synthesized by the carbodiimide coupling reaction [ 31 ]. The reaction mixtures (7.5 mL) containing 5 mM methyl ester derivatives of histidine or methylated histidine, 15 mM BOC-β-alanine or BOC-GABA, 10 mM HOBt, 200 mM 2-morpholinoethanesulfonic acid buffer (pH 7.0), and 15 mM EDC were stirred for 2 h on ice, followed by incubation for another 12 h at room temperature (25 °C). After the removal of the solvent in vacuo, the synthesized compound was purified by HPLC using a Scherzo SS-C18 column (10.0 × 150 mm; Imtakt, Kyoto, Japan) and a discontinuous gradient of solvent A (water containing 0.1% formic acid) and solvent B (water containing 50% acetonitrile and 100 mM ammonium formate) (0% B at 0 min; 50% B at 0.1 min; 50% B at 5 min; 90% B at 10 min) at a flow rate of 3.0 mL/min. The elution was monitored by absorbance at 220 nm. The eluate containing the synthesized compound was collected, an equal amount of 1 M NaOH was added to the collected eluate to deprotect methyl ester groups, and the mixture was stirred for 15 min at room temperature (25 °C). Thereafter, the mixture was acidified by adding an equal volume of 6 M HCl and further incubated with an equal volume of 6 M HCl for 90 min at room temperature (25 °C) to deprotect BOC groups. The crude IDP was purified by DOWEX™ 50WX8 cation exchange resin (5 mL; Wako Pure Chemical Corporation, Osaka, Japan) and HPLC using a Scherzo SS-C18 column (10.0 × 150 mm; Imtakt, Kyoto, Japan) under the following conditions: a discontinuous gradient of solvent A (water containing 0.1% formic acid) and solvent B (water containing 50% acetonitrile and 100 mM ammonium formate) (0% B at 0 min; 30% B at 0.1 min; 70% B at 20 min; 100% B at 20.1 min) at a flow rate of 3.0 mL/min. The elution was monitored by absorbance at 220 nm. The purity of synthesized IDPs were analyzed by HPLC-photodiode array (PDA) analysis ( Supplementary Figure S1 ). To determine the concentrations of synthesized IDPs and stable isotope labeled IDPs, they were hydrolyzed with 6 N HCl for 9 h at 110 °C, and the concentrations of β-alanine or GABA were quantified by HPLC-ESI-MS/MS. Experimental procedures described in this section are shown in Supplementary Figure S2
To synthesize methyl ester derivatives of histidine and its methylated analog, 50 mg of histidine or its methylated analog was suspended in methanol (1 mL). Thionyl chloride (200 µL) was added dropwise and the mixture was stirred for 3 h at 50 °C. The solvent was removed in vacuo, resulting in methyl ester derivatives. Next, to prepare BOC derivatives of [ 13 C 3 , 15 N]β-alanine, the reaction mixtures of dioxane (250 µL) and 0.5 mM NaOH (250 µL) containing 20 mM [ 13 C 3 , 15 N]β-alanine and 60 mM di- tert -butyl dicarbonate were stirred for 90 min at room temperature (25 °C). The samples were then dried in vacuo and purified by HPLC (JASCO, Tokyo, Japan) under the following conditions: a Mightysil RP-18 GP column (6.0 × 150 mm; Kanto Chemical, Tokyo, Japan) using a linear gradient of solvent A (water) and solvent B (methanol) (0% B at 0 min; 50% B at 16 min) at a flow rate of 3.0 mL/min. The elution was monitored by UV absorbance at 190 nm. After the concentration of the eluate containing BOC derivatives in vacuo, the concentration of BOC-[ 13 C 3 , 15 N]β-alanine was determined by HPLC-UV using the standard curve.
Fresh beef (loin, Australia), pork (loin, Japan), chicken (breast, Japan), and minke whale (red meat, Southern Ocean) samples were purchased from various suppliers around the world. Di- tert -butyl dicarbonate and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide monohydrochloride (EDC) were obtained from Peptide Institute (Osaka, Japan). β-Alanine, γ-aminobutyric acid, and l -ascorbic acid were obtained from Nacalai Tesque (Kyoto, Japan). N - tert -Butoxycarbonyl (BOC)-β-alanine, BOC-GABA, l -histidine methyl ester dihydrochloride, 1-hydroxybenzotriazole monohydrate (HOBt), and 2,2-diphenyl-1-picrylhydrazyl (DPPH) were obtained from Tokyo Chemical Industry (Tokyo, Japan). 1-Methyl- l -histidine and [ 13 C 3 , 15 N]β-alanine were obtained from Sigma-Aldrich (St. Louis, MO, USA). 3-Methyl- l -histidine was obtained from Bachem AG (Torrance, CA, USA). Copper(II) sulfate, 5-hydrate was obtained from Kishida Chemical (Osaka, Japan). Trolox was obtained from Merck (Darmstadt, Germany). All other chemicals and reagents were obtained from common suppliers and were of the highest grade commercially available.
The quantitative analyses of IDPs and 2-oxo-IDPs are summarized in Table 4 . All IDPs were detected in all samples in this study, except for homoanserine in whale meat. Carnosine was a predominant IDP in beef and pork, whereas anserine and balenine were the major IDPs in chicken and whale meat, respectively. Homocarnosine and homoanserine were also detected, although the levels of these IDPs were considerably lower than those of major IDPs. The total concentrations of IDPs in beef, pork, chicken, and whale meat were 250, 340, 300, and 690 nmol/mg protein, respectively.
To determine the LOD and LOQ of IDPs, standard calibration curves were generated in the concentration range of 1–1000 fmol. Supplementary Figure S6A shows the regression equation for the calibration curve of IDPs and revealed that IDPs exhibited good linearity (= 0.999). Similarly, the standard calibration curves of 2-oxo-IDPs were constructed in same concentration range and showed good linearity (= 0.999) ( Supplementary Figure S6B ). An analysis of the signal-to-noise ratio revealed that the LOQ values of IDPs and 2-oxo-IDPs were in range of 240.1–2561 ng/mL by this method ( Table 3 ), corresponding to in range of 10–100 fmol. Furthermore, the lowest LOD was 3 fmol (72.03 ng/mL) for IDPs and 2-oxo-IDPs.
To avoid the co-elution of isomers, we examined various columns and elution condition, and optimized the HPLC condition for the enough separation of dipeptides. Retention times of the peaks of balenine, anserine, homoanserine, carnosine, and homocarnosine were 8.31, 9.29, 9.36, 9.89, and 9.98 min, respectively ( Figure 4 A). Another peak was detected at the same retention time as that of balenine in the chromatogram of anserine and this peak may be attributed to common precursor ion shared between balenine and anserine (241.3 → 109.1). However, these peaks were adequately separated in the HPLC system and we concluded that these dipeptides were distinguishable on the chromatogram. 2-Oxo-IDPs were also separated under optimized HPLC conditions, and the peaks of 2-oxo-balenine, 2-oxo-anserine, 2-oxo-homoanserine, 2-oxo-carnosine, and 2-oxo-homocarnosine were sufficiently separated and detected at retention times of 3.85, 4.40, 4.57, 4.73, and 4.92 min, respectively ( Figure 4 B). Although a minor peak was detected in the chromatogram of 2-oxo-anserine at the same retention time as that of 2-oxo-balenine, each peak of 2-oxo-IDPs was sufficiently separated.
Furthermore, to evaluate the chemical property of 2-oxo-IDPs, we performed a DPPH radical scavenging assay. As shown in Figure 3 , all of the 2-oxo-IDPs exhibited a greater antioxidant capacity than that of the corresponding precursor; IDPs showed little or no antioxidant capacity despite being used at a higher concentration (i.e., 200–1000 μM) than that of 2-oxo-IDPs (i.e., 20–40 μM). This is the first evidence of the potent antioxidant capacity of 2-oxo-baleinine and 2-oxo-homocarnosine.
Next, we synthesized 2-oxo-IDPs by the copper/ascorbate system as described previously [ 7 ], with slight modifications. Figure 2 A shows the result of the MS/MS analysis of 2-oxo-balenine; major product ions at210.1, 169.1, 123.1, 89.0, and 72.0 were detected. Combined with the results of collision-induced dissociation of stable isotope-labeled 2-oxo-balenine ( Supplementary Figure S5A,B ), the losses of CHNHand OH were speculated to result in the formation of the product ion at210.1, the product ions at169.1 and 123.1 were expected to originate from 2-oxo-1-methyl-histidine, and the product ions at89.0 and 72.0 were derived from the β-alanine moiety. These product ions were identified as shown in Figure 2 B. Similarly, MS/MS spectra of other 2-oxo-IDPs are shown in Figure 2 C–F, and the assignment of product ions was performed ( Supplementary Figure S5C–F ), revealing the successful synthesis of 2-oxo-IDPs. The MRM parameters for the quantification of 2-oxo-IDPs are shown in Table 2
Although carnosine and anserine are commercially available from some suppliers, other IDPs (i.e., balenine, homocarnosine, and homoanserine) and stable isotope-labeled IDPs are seldom available. Therefore, we prepared IDPs and stable isotope-labeled IDPs by chemical synthesis using the carbodiimide coupling reaction. The synthesis of IDPs was confirmed by LC-UV detection and HPLC-ESI-MS/MS. Figure 1 A shows the result of the MS/MS analysis of balenine. Collision-induced dissociation of balenine revealed major product ions at224.1, 170.1, 124.1, and 109.1. We speculated that the loss of NHresulted in the formation of the product ion at224.1, and the product ions at170.1, 124.1, and 109.1 originated from a 1-methyl-histidine moiety. These product ions were identified as shown in Figure 1 B. The assignment of these product ions was supported by the observation that the collision-induced dissociation of stable isotope-labeled balenine, containing [N]β-alanine, produced relevant product ions at227.1, 170.1, 124.1, and 109.1 ( Supplementary Figure S4A,B ). Similarly, the MS/MS spectra of homocarnosine and homoanserine are shown in Figure 1 C,D, and the assignment of product ions was performed ( Supplementary Figure S4C,D ). Furthermore, carnosine and anserine were subjected to collision-induced dissociation to compare the MS/MS spectra, and the results are shown in Figure 1 E,F. Carnosine and homocarnosine produced common product ions at156.1, 110.1, and 95.1, which can be attributed to the histidine moiety. Similarly, anserine and homoanserine generated common product ions at170.1, 126.1, and 109.1, expected to originate from 3-methyl-histidine. The characteristic collision-induced dissociation and the assignment of product ion revealed that the IDPs were successfully synthesized. Next, we determined the multiple reaction monitoring (MRM) parameters for the quantification of IDPs based on MS/MS results, and the collision energies were optimized to produce the highest signal intensities of product ions ( Table 1 ).
4. Discussion
m/z 241; however, MS/MS spectra of the fragment ions from these IDPs differed slightly. Turecek et al. reported that the precursor ion of 1-methyl- l - histidine (component of balenine), which appeared at m/z 170, underwent the dominant loss of H 2 O + CO to form a fragment ion at m/z 124; in contrast, 3-methyl- l - histidine (component of anserine) showed a loss of CO 2 to form a fragment ion at m/z 126 as important primary dissociations [ m/z 124, whereas those of anserine produced the highest ion peak at m/z 109 [ In the current study, we synthesized IDPs for the preparation of 2-oxo-IDPs and internal standards for accurate MS analyses. Although carnosine and anserine are commercially available from suppliers, the availability of other IDPs (e.g., balenine, homocarnosine, and homoanserine) and stable isotope-labeled IDPs is limited. Here, we synthesized several types of IDPs, including stable isotope-labeled balenine, by the carbodiimide coupling reaction. By HPLC-ESI-MS/MS, anserine, balenine, and homocarnosine were detected as precursor ions at241; however, MS/MS spectra of the fragment ions from these IDPs differed slightly. Turecek et al. reported that the precursor ion of 1-methyl-histidine (component of balenine), which appeared at170, underwent the dominant loss of HO + CO to form a fragment ion at124; in contrast, 3-methyl-histidine (component of anserine) showed a loss of COto form a fragment ion at126 as important primary dissociations [ 32 ]. They also demonstrated that these differences in dissociation were related to a difference in the methylation site in the imidazole ring. Uenoyama et al. reported that MS/MS analyses of balenine produced the highest ion peak at124, whereas those of anserine produced the highest ion peak at109 [ 33 ]. These findings are consistent with our observations, indicating that the IDPs were successfully synthesized. We also prepared 2-oxo-IDPs and stable isotope-labeled 2-oxo-balenine by the copper/ascorbate oxidation system [ 7 ], using synthesized IDPs and stable isotope-labeled IDPs, respectively, as starting materials. The results of MS/MS analyses were consistent with previous results [ 7 23 ], suggesting the successful synthesis of 2-oxo-IDPs. Consequently, we successfully prepared five IDPs, five 2-oxo-IDPs, and their stable isotope-labeled compounds. The use of these compounds as standards enabled their quantitative detection by HPLC-ESI-MS/MS.
24,24, HPLC-ESI-MS/MS has been used for the detection and quantitation of IDPs [ 30 ]. Macia et al. developed a LC-MS method for the rapid, sensitive, and selective determination of carnosine and anserine in samples containing complex matrices [ 34 ]. However, matrix effects, such as ion suppression, must be considered during HPLC-ESI-MS/MS-based detection to achieve reliable analytical data [ 35 ]. To correct for this effect, an isotope dilution method has been used for the exact quantitative MS analysis of carnosine [ 31 36 ], anserine [ 31 37 ], homocarnosine [ 38 ], and homoanserine [ 23 ]. However, no study has used isotope-labeled balenine. The application of HPLC-ESI-MS/MS coupled with a stable isotope dilution method for 2-oxo-IDPs has only been demonstrated by our previous reports for the detection of 2-oxo-carnosine [ 7 25 ], 2-oxo-anserine [ 7 25 ], 2-oxo-homocarnosine [ 7 ], and 2-oxo-homoanserine [ 23 ]. However, no study has demonstrated about 2-oxo-balenine. In this study, we developed quantitative HPLC-ESI-MS/MS methods using isotope-labeled balenine and 2-oxo-balenine. To the best of our knowledge, this is the first demonstration of the quantitative analysis of balenine and 2-oxo-balenine using HPLC-ESI-MS/MS coupled with a stable isotope dilution method. Together with our previous reports, it is now possible to detect IDPs and 2-oxo-IDPs specifically and quantitatively by using these two different HPLC programs followed by MS/MS analysis, respectively. Moreover, we demonstrated the highly sensitive and selective detection of IDPs and 2-oxo-IDPs in MRM mode using HPLC-ESI-MS/MS and achieved detection at the level of 3 fmol. Jozanović et al. summarized recent analytical methods for the detection of carnosine, reporting LOD values in the range of 4.5–7000 fmol [ 39 ]. The LOD of our current method is comparable to or lower than those of other analytical techniques. Therefore, the newly established method using HPLC-ESI-MSMS combined with stable isotope dilution enabled the highly sensitive and exact detection of five 2-oxo-IDPs.
33, We determined the contents of five IDPs and five 2-oxo-IDPs in beef, pork, chicken, and whale meat samples by HPLC-ESI-MSMS coupled with a stable isotope dilution method. As shown in Table 4 , we successfully detected five IDPs from all meat samples used in this study, and the contents and proportion of IDPs in various sample types were approximately consistent with those of previous reports [ 6 40 ]. Various studies have evaluated IDP contents in meat; however, many of them focused only on the content of predominant IDPs. In fact, only a few studies have reported the content of homocarnosine in meat [ 41 42 ], and studies of the homoanserine content in meat are lacking. We successfully detected homoanserine in meat, including beef, pork, and chicken, for the first time using the newly developed method. These results support the use of our quantitative method for IDP detection and the evaluation of minor constituents in meat. Moreover, analytical methods based on stable isotope dilution provide reliable data on the absolute amounts of several IDPs, providing a basis for comparative analyses of IDP compositions in meat.
We successfully identified several 2-oxo-IDPs in meat in a single measurement. Notably, this is the first evidence for the endogenous production of 2-oxo-balenine, which was detected in not only whale but also in other meat types. The total amount of 2-oxo-IDPs was 100–280 pmol/mg protein and the percentage of 2-oxo-IDPs to IDPs was 0.015–0.11%. We have previously reported 2-oxo-IDP contents in meat of 48–340 pmol/mg protein, accounting for 0.027–0.26% of the total IDP contents [ 24 ], in concordance with the results of the current study. We also showed that the predominant types of 2-oxo-IDPs were consistent with the predominant IDPs, indicating that the amounts of 2-oxo-IDPs were correlated with those of IDPs in meat. This observation is in line with previous results [ 7 23 ]. Thus, the highly sensitive and specific detection method developed here provides precise insight into the content and distribution of 2-oxo-IDPs.