1. Introduction
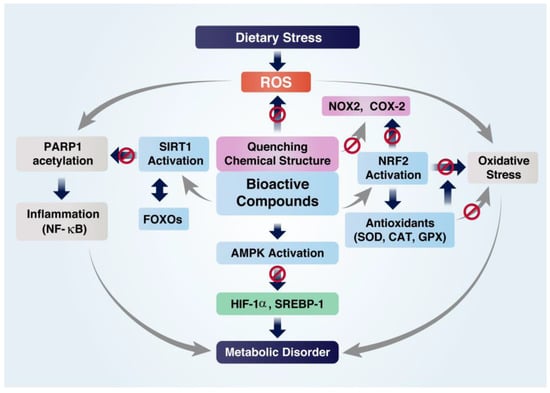
Bioactive compounds (BCs) are nutritional agents with diverse potentials exhibiting anti-inflammation, anti-oxidative stress, and anti-metabolic syndrome. The ability of BCs to modulate biological and physiological conditions may derive from their chemical and biological structures for favorable bioavailability and biochemical function [ 1 2 ]. The unique chemical structures of BCs enable them to effectively quench reactive oxygen species (ROS) for redox balance [ 2 ]. BCs have been demonstrated to control the expression of target genes and proteins via regulating epigenetic modifications, including the regulation of the histone acetylation state [ 3 4 ]. In redox signaling, inflammation, oxidative stress, and metabolism are considered electron-transfer processes reliant on the electron transport chain (ETC) [ 5 6 ]. The transfer of electrons to the final acceptor of the oxygen molecule is required for ATP synthesis. During several processes modulated by electron donors and acceptors, redox balance and stability are directly related to the metabolism and immune responses [ 7 8 ]. A sustainable and stable redox balance is essential for maintaining physiological pathways within the cells [ 7 ].
The function of BCs influences epigenetic changes that affect DNA repair and cell proliferation through the deacetylation of histones or non-histone proteins [ 9 10 ]. Depending on the redox environments, the post-translational modifications of histones are available in the N-terminal of core histone protein [ 10 11 ]. The alteration in histone acetylation is controlled by the opposing actions of histone acetyltransferases (HATs) and histone deacetylases (HDACs) [ 4 9 ]. The modulation of acetyl moieties can affect metabolic reprogramming and immune response. The hyperacetylation of histones has been reported to result in DNA damage, whereas hyperacetylated non-histone proteins increase ubiquitination [ 1 3 ]. Hypoacetylation with HDACs led to the exacerbation of diseases, while significant decreases in HDAC activity were found in patients with several diseases [ 11 ]. Histone deacetylation targeting the chromatin remodeling and its mobility resulted in the control of genomic binding for DNA recombination and access for histone-DNA interactions [ 12 ]. The coordination of epigenetic regulators is likely based on the redox state, as moieties’ accessing and binding features depend on the electric charge.
15,18, The dietary stress-induced change of the redox state causes a highly oxidative condition due to the excessive generation and accumulation of ROS, leading to oxidative stress, inflammation, and metabolic dysfunctions [ 2 13 ]. The oxidation state can be altered by supplementing BCs with the ability to scavenge ROS and electrons and activate sirtuin 1 (SIRT1) and nuclear factor erythroid 2–related factor 2 (NRF2), a master regulator of anti-oxidative responses [ 5 6 ]. Dietary stress, i.e., excessive alcohol consumption and a high-fat or high-glycemic diet, can cause alterations in glucose and lipid metabolism, leading to excessive fat accumulation and the production of ROS and inflammatory cytokines [ 14 16 ]. These metabolic signaling changes can alter cancers, liver diseases, diabetes, pancreatitis, and atherosclerosis [ 17 19 ]. The dietary stress triggers toll-like receptors (TLRs) and promotes the activation of nuclear factor-κB (NF-κB) or activator protein 1 (AP-1) by acetylation, increasing inflammation [ 14 16 ].
21,30,35, SIRT1 and NRF2 are considered prime targets for edible BCs [ 20 22 ]. SIRT1, a class III HDAC, is encoded in a nuclear-located gene with the largest N-terminal and C-terminal domains among its seven families [ 11 23 ]. The activation of SIRT1 adjusts the redox state to prevent inflammation, oxidative stress, and disorders by facilitating the positive feedback loop with NRF2 signaling [ 24 ]. SIRT1 is reported to promote the formation of the SIRT1-NF-κB or SIRT1-AP-1 axis and inactivate target transcription factors by deacetylation [ 25 26 ]. Cellular stress caused by excessive alcohol, high-fat, or high-glycemic diet may induce the inhibition of SIRT1 by the inhibitory protein, p53-acetylated peptides, forming a pseudo-substrate for SIRT1 inactivation [ 27 28 ]. However, supplementing BCs favors the interaction with SIRT1 to activate NAD+-regulatory elements in the C-terminal domain, resulting in the activation of SIRT1 [ 29 31 ]. In mouse liver, high-fat diet-induced p53 acetylation and transcriptional activity resulted in a lower level of SIRT1 [ 32 33 ]. However, the activation of SIRT1 by supplementing BCs repressed p53 activity by deacetylating it [ 27 28 ]. The deacetylation ability of SIRT1 activates farnesoid X Receptor (FXR) to promote the transcription of the small heterodimer partner (SHP), resulting in the repression of p53 and an increase in SIRT1 expression [ 34 36 ].
The present review summarized the current studies on the BCs exhibiting protective effects against inflammation, oxidative stress, and metabolic dysfunction by regulating the cellular redox balance and acetylation states of histone and non-histone proteins. Excessive alcohol, high-fat, or high-glycemic diet was considered dietary stress for the induction of the immune system and metabolic disorders. The results of this work can be utilized to provide a platform for developing therapeutic interventions by employing BCs.