The purpose of this study was to determine the effects of a 14-day intake of whole freeze-dried WB powder on the FAT-ox rate in healthy males during a 40 min submaximal cycling bout. The objectives were to determine (1) the rate of FAT-ox during submaximal exercise using respiratory exchange ratio (RER) and substrate oxidation (FAT-ox and CHO-ox) rates and (2) the response of plasma lactate and urinary F2-isoprostanes, creatinine, and free/total carnitine during submaximal exercise. It was hypothesized that consuming 375 g/d of anthocyanins from 25 g/day of freeze-dried wild blueberry powder would result in a greater rate of FAT-ox during cycling at 65% of VO 2peak for 40 min.
Dietary interventions aiming to maximize FAT-ox during exercise through anthocyanin-rich fruit are limited to three studies that supplemented with New Zealand blackcurrants (NZBC) [ 23 25 ]. Cook et al. (2015) demonstrated that 105 mg of anthocyanin from 300 mg of NZBC extract for seven days resulted in a 27% increased rate of FAT-ox via expired air sampling calculations in trained male cyclists at 65% of VO. In their 2017 study, Cook et al. documented a significant dose effect for average FAT-ox increase of 21.5% and 24.1% from 0 mg/day to 600 mg (210 mg of anthocyanin) and 900 mg/day (315 mg of anthocyanin) of NZBC extract, respectively, in trained male cyclists at 65% of VO. Further research with trained female cyclists consuming 210 mg of anthocyanins from 600 mg of NZBC extract for seven days also resulted in a 27% increase in FAT-ox via expired air calculations and a significant increase in non-esterified fatty acids by blood collection [ 25 ]. Wild blueberries (WB) and NZBC contain similar amounts of anthocyanins [ 26 ], although WBs are more widely available in the United States. Research on the effects of WB on FA mobilization and oxidation during exercise has yet to be conducted, as the majority of studies have focused on the health benefits of WB [ 27 28 ].
In addition to various physical training programs, athletes are turning to dietary strategies to enhance performance, such as incorporating anthocyanins in their diet. Consumption of anthocyanins [ 10 ] has been favorable due to their anti-inflammatory and antioxidant functions [ 11 12 ]. Higher amounts of anthocyanins can be found in fruits with red, blue, and purple hues, such as wild blueberries, tart cherries, black currants, red and black grapes, and pomegranates [ 13 ]. Recent evidence suggests that the consumption of anthocyanin-rich fruits may also improve lipid mobilization and oxidation [ 13 17 ]. Although the specific mechanism of action is not yet determined, possible explanations include an increase in enzymatic action reducing fat absorption [ 18 ], increases in AMPK and CPT-1 activity in whole-body FAT-ox [ 19 20 ], increased transportation of FA into mitochondria [ 20 ], and increased peripheral blood flow to working muscles [ 21 ], thus increasing lipolysis and FAT-ox [ 2 22 ].
Attaining a higher rate of fat oxidation (FAT-ox) has various cardiometabolic benefits and when attained during exercise, endurance-based exercise performance may be enhanced. Adipose tissue, plasma triglycerides (TG), and intramuscular TGs supply fatty acids (FA) that are predominantly used for energy production during lower-intensity exercise [ 1 2 ]. As exercise intensity increases, the rate of FAT-ox decreases and cannot meet fuel demands, leading to a greater reliance on glycogen sources [ 1 7 ]. Unfortunately, sustained reliance on glycogen can result in glycogen depletion and lead to fatigue and decreased exercise capacity. Maximizing fat mobilization and oxidation and slowing glycogen depletion may improve performance at various exercise intensities [ 8 9 ].
2. Materials and Methods
2peak ≥ 45 mL/kg/min. All methods were approved by the Institutional Review Board at California Polytechnic University, Humboldt, in accordance with the Declaration of Helsinki. Eleven healthy aerobically trained males (aged 26.55 ± 7.95) participated in the study (see Table 1 for descriptive statistics). Previous work examining FAT-ox rates following anthocyanin-rich fruit consumption was the basis for calculating a G *Power analysis set at a power of 80% and an α of.05 indicated a sample of 12 participants to detect a difference of 2–3% (G*Power [ 25 ]). Participants were included if they were: ≥18 years old; non-tobacco users; waist circumference < 102 cm [ 29 ]; free of any cardiovascular, metabolic, respiratory, and orthopedic conditions; not taking blood pressure and/or cholesterol-lowering medications; and not following a high fat/low carbohydrate diet. All participants engaged in moderate-intensity cardiorespiratory exercise at least three days a week for ≥ 30 min for the past two years and had a VO≥ 45 mL/kg/min. All methods were approved by the Institutional Review Board at California Polytechnic University, Humboldt, in accordance with the Declaration of Helsinki.
2peak protocol to determine eligibility followed by a familiarization trial. Participants were then instructed to follow the C diet for two weeks before returning for visit 2. This was a non-randomized, quasi-experimental, free-living trial. Participants were required to visit the Human Performance Laboratory at California Polytechnic University, Humboldt, at the same time (7:00 a.m.) on three separate visits (see Figure 1 ) over the span of four weeks and to complete two diet conditions: diet washout (C) and WB intervention. During visit 1, participants completed the consent process, health history, and diet and exercise questionnaires; had anthropometric measures taken; and then completed a VOprotocol to determine eligibility followed by a familiarization trial. Participants were then instructed to follow the C diet for two weeks before returning for visit 2.
Two weeks after visit 1, participants returned for visit 2 at 7 a.m. after a 12 h fast following a mixed meal the night before containing carbohydrates, proteins, and fats consumed before 7 p.m. Upon consuming the meal, participants could continue to consume water in the evening and morning prior to the session. Participants were instructed to avoid strenuous exercise 48 h prior to visit 2; avoid alcohol within 24 h; avoid caffeine, aspirin, and NSAIDs the morning of; and collect and return their first-morning urine. During visit 2, once participants were seated and ready to begin, they were given a finger stick puncture to collect 300 microliters of capillary blood for later analysis. Once blood was collected, participants began warming up on the bike for 15 min, wherein the first 5 min were spent working up to 45% of VO 2peak , and then, for the next 10 min, they slowly increased intensity until reaching 65% of VO 2peak , which they maintained for 40 min. Immediately before, after, and every 10 min of the 40 min exercise protocol, 5–10 microliters of blood via finger stick puncture were collected to measure lactate. Immediately following the exercise protocol, participants were given another finger stick puncture to collect an additional 300 microliters of capillary blood for later analysis. Participants were allowed to cool down on the bike at their comfort level until they were ready to get off. As soon as they were able, following the exercise protocol, participants collected a second urine sample. All participants were able to collect their second sample within 15 min of completing the protocol. Following visit 2, participants were instructed to consume the WB intervention twice a day for the next two consecutive weeks while maintaining the diet washout. The third and final visit occurred two weeks after the second session when participants repeated the protocol and preparatory instructions from visit 2. Participants were instructed by trained staff to record dietary intake and physical activity during both the diet washout and WB consumption periods. Fluid intake was individualized on the basis of body weight for each subject during the two days prior to visits 2 and 3.
Diet washout: During the washout period, participants were given a list of foods to avoid and/or limit for two weeks before their second visit. This included commonly consumed foods that contain large amounts of anthocyanins such as fruits and vegetables with blue, purple, and red colors, along with red wine. Participants were also asked to avoid green tea for the remainder of the study as green tea has been shown to increase [ 30 ]. The purpose of the washout period was to ensure that any differences seen in FAT-ox the measured variables between the C (pre-supplementation) and WB conditions could be attributed to the WBs. The participants were instructed to follow a similar diet of no additional anthocyanin intake for the remainder of the study.
26, WB diet: Freeze-dried whole WB powder was donated by The Wild Blueberry Association in Maine containing berries from the 2017 crop. The process of freeze-drying the berry results in the least reduction in polyphenol content [ 31 ]. Participants were provided with a jar and food-grade blender ball and instructed to mix 12.5 g of the powder in 125 mL of water twice a day for two weeks. The 25 g of total daily intervention intake is equivalent to one cup of raw fruit providing 375 mg of anthocyanins and has been documented as an effective dose to decrease levels of oxidized DNA bases and increase the resistance to DNA damage [ 27 ] and is higher than the effective doses of NZBC found to increase FAT-ox in exercise [ 25 27 ]. The WB intervention provides 101.5 kcals, 92 g CHO, 2.15 g protein, and 15.88 g of fiber per day. Further instructions included avoiding food intake, especially dairy products, 30 min before and after WB consumption. Participants were also asked to consume the WB intervention in the mornings and evenings with a minimum of 8 h apart. Lastly, it was recommended that participants consume the WB intervention at the same time of day throughout the entire two weeks.
2peak was determined using incremental stage protocol until volitional fatigue on a Velotron cycle ergometer (RacerMate, Seattle, WA, USA) [ 2peak protocol, participants cooled down on the bike for 5 min before completing a familiarization trial consisting of 15 min of cycling at 65% of VO 2peak . Given that fat is the primary fuel source during low-to-moderate intensity exercise, peaking at about 60–65% of VO 2max , after about 15–20 min, with glucose becoming the primary fuel source as intensity increases (≥85% VO 2max ) [ 2peak and sustained steady-state cycling for 40 min. Heart rate (HR), power output (measured in watts [W]), cadence, and rate of perceived exertion (RPE) using the Borg scale was measured at baseline and at 10 min intervals throughout the 40 min of cycling. VOwas determined using incremental stage protocol until volitional fatigue on a Velotron cycle ergometer (RacerMate, Seattle, WA, USA) [ 32 ]. Briefly, the protocol included a 3 min warm-up at zero watts followed by 25-watt increases every minute. After completion of the VOprotocol, participants cooled down on the bike for 5 min before completing a familiarization trial consisting of 15 min of cycling at 65% of VO. Given that fat is the primary fuel source during low-to-moderate intensity exercise, peaking at about 60–65% of VO, after about 15–20 min, with glucose becoming the primary fuel source as intensity increases (≥85% VO) [ 33 ], the exercise testing protocol consisted of a 15 min warm-up to reach 65% of their VOand sustained steady-state cycling for 40 min. Heart rate (HR), power output (measured in watts [W]), cadence, and rate of perceived exertion (RPE) using the Borg scale was measured at baseline and at 10 min intervals throughout the 40 min of cycling.
Body composition was estimated via seven site skinfold measures, namely, the tricep, subscapular, chest, midaxillary, abdomen, suprailiac, and thigh on the right side of the body using Lange skinfold calipers (Beta Technology, Santa Cruz, CA, USA). Measurements were completed in duplicate and averaged. Triplicate measures were taken when the first two skinfold measurements had a difference >2 mm [ 29 ]. Skinfold averages for each site were summed, and body density was calculated using the seven-site formula for men and was converted into body fat percentage using the Siri equation [ 29 ]. Waist circumference was measured at the upper ridge of the iliac crest using a Baseline measurement tape with Gulick attachment (Fabrication Enterprises Inc., Elmsford, NY, USA). When duplicate WC measures were > 5 mm, a triplicate measure was taken. Skinfold and WC measurements were taken at each visit to the lab to monitor changes in body composition and weight distribution.
During the C and WB intervention periods, participants were instructed to maintain and record current training regimens and daily physical activity in detail including type, duration, and intensity. Participants were also instructed to maintain current diet patterns, aside from foods designated to avoid or limit during the C and WB intervention periods, and record their diet (type, brand, preparation method, recipes, and amount of food and beverage consumed) for the three days prior to both visit 1 and visit 2. Participants were encouraged to submit pictures of their meals with their hand next to the food for serving size reference. Self-reported dietary intake and physical activity records were collected and reviewed with participants at the start of each visit. Diet records were then entered into Food Processor version 8.0 (ESHA, Salem, OR, USA) and analyzed by trained research staff; physical activity records were analyzed on the basis of total minutes/week of anaerobic and aerobic activity. Participants reported compliance with the supplementation protocol, diet, and physical activity instructions.
2 , VCO 2 , ventilation (V E ), and RER were assessed with a metabolic cart (TrueOne 2400 metabolic cart, PARVO Medics, Sandy, UT, USA) [ Fat oxidation (g/min) = 1.695 × V · O 2 − 1.701 × V · CO 2 Carbohydrate oxidation (g/min) = 4.210 × V · CO 2 − 2.962 × V · O 2 The primary measures included FAT-ox and CHO-ox rates using expired gases, capillary whole blood FA and glycerol, with F2-isoprostanes (≈50 mL), creatinine, and free/total carnitine (≈250 µL) via urine collection. VO, VCO, ventilation (V), and RER were assessed with a metabolic cart (TrueOne 2400 metabolic cart, PARVO Medics, Sandy, UT, USA) [ 34 ], while rates of whole-body FAT-ox and CHO-ox were determined using the following mathematical equations with the assumption that protein contribution is minor [ 35 ]:
Both fingerstick whole blood and urine samples were collected pre- and post-exercise sessions and stored at −80 °C for later analysis. Glycerol and FFA were analyzed using Cayman Chemical (Ann Arbor, MI) ELISA kits at the Core Lab at Cal Poly Humboldt. F2-isoprostanes, creatinine, and free/total carnitine were analyzed using LC–MS by the Linus Pauling Institute at Oregon State University. Additional measures included lactate taken via finger puncture using the Lactate Plus Meter (Nova Biomedical, Waltham, MA, USA).
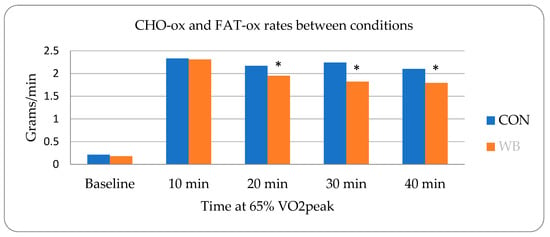
Statistical analysis was performed with JMP statistical software version 15.0 (SAS institute, Cary, NC, USA). Repeated measures analysis of variance (ANOVA) with post hoc Tukey HSD tests were used to determine differences between the dependent variables (FAT-ox, CHO-ox, lactate, urinary F2-isoprostanes, blood FA, glycerol, creatinine, free/total carnitine, RER, V e , heart rate, power output, cadence, and RPE) before, during, and after 40 min of cycling using condition (WB vs. CON) by time-point (pre vs. post or minutes 0, 10, 20, 30, 40). Mauchley’s test of sphericity determined the homogeneity of data, and Greenhouse–Geiser adjustments were used for violations. Differences in physical activity and food intake between the two weeks before each condition were analyzed using one-way ANOVA with pooled t-tests assuming equal variance. Mauchley’s test of sphericity determined homogeneity of data, and Greenhouse–Geiser adjustments were used for violations.