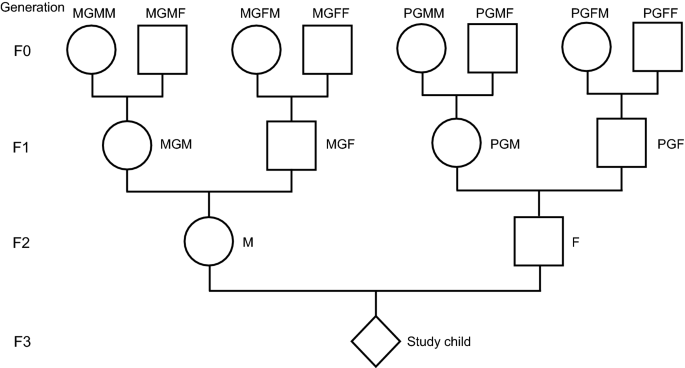
The ALSPAC population
ALSPAC was designed to assess ways in which aspects of the environment and genes of individuals may interact to result in benefits and disadvantages to health and development14. It started during the pregnancies of women who were resident in a predefined area (that part of Avon that was within the South-West Regional Health Authority) and had an expected date of delivery between 1st April 1991 and 31st December 1992. Eligible women were contacted as early in pregnancy as feasible. They and their offspring were followed throughout pregnancy and then through childhood, adolescence and into adulthood. The collection of information is continuing. Data were collected using a variety of methods including questionnaires completed by the mothers, their partners and their offspring; analysis of biological samples; linkage to standard data sets, and hands-on examinations.
The initial number of pregnancies enrolled is 14,541 (for these at least one questionnaire has been returned or a “Children in Focus” clinic had been attended by 19/07/99). Of these initial pregnancies, there was a total of 14,676 fetuses, resulting in 14,062 live births and 13,988 children who were alive at 1 year of age. When the oldest children were approximately 7 years of age, an attempt was made to bolster the initial sample with eligible cases who had failed to join the study originally. As a result, when considering variables collected from the age of seven onwards (and potentially abstracted from obstetric notes) there are data available for more than the 14,541 pregnancies mentioned above. The number of new pregnancies not in the initial sample (known as Phase I enrolment) that are currently represented on the built files and reflecting enrolment status at the age of 24 is 913 (456, 262 and 195 recruited during Phases II, III and IV respectively), resulting in an additional 913 children being enrolled. The phases of enrolment are described in more detail in the cohort profile paper and its update15,16,17. The total sample size for analyses using any data collected after the age of seven is therefore 15,454 pregnancies, resulting in 15,589 fetuses. Of these 14,901 were alive at 1 year of age. Study data were collected and managed using REDCap electronic data capture tools hosted at the University of Bristol. REDCap (Research Electronic Data Capture) is a secure, web-based software platform designed to support data capture for research studies18. Please note that the study website contains details of all the data available through a fully searchable data dictionary and variable search tool: [https://www.bristol.ac.uk/alspac/researchers/our-data/]. Ethical approval for the study was obtained from the ALSPAC Ethics and Law Committee (ALEC; IRB00003312) and the Local Research Ethics Committees. Detailed information on the ways in which confidentiality of the cohort is maintained may be found on the study website: http://www.bristol.ac.uk/alspac/researchers/research-ethics/.
All methods were performed in accordance with the relevant guidelines and regulations. Informed consent for the use of data collected via questionnaires and clinics was obtained from participants following the recommendations of the ALSPAC Ethics and Law Committee at the time19.
As part of the original data collected during pregnancy, the questionnaires sent to the study mother and her partner (usually the father of the study child) included details of their childhood and adolescence, including the age at which they had started smoking regularly, together with other information on their smoking habits, and those of their parents (i.e., the study child’s grandparents). However, the smoking habits of these grandparents did not include details as to their ages when they had started smoking. Consequently, a recent endeavour has resulted in the sending of new questionnaires to those biological parents with whom the study was still in contact, to obtain further information on their parents and grandparents, including the age at which they had started smoking regularly. Questionnaires were mainly sent online, but for those who preferred paper alternatives, paper questionnaires were posted to them. Full details of the methodology and the questions asked can be found elsewhere20.
The outcomes
Total fat and lean mass were estimated with the use of a Lunar Prodigy DXA scanner (GE Medical Systems Lunar, Madison, WI). The scans were visually inspected and realigned when necessary. For the present study we have used the measurements of fat and lean mass collected at face-to-face clinics at the ages of 17 (approximating to the end of puberty) and 24 years (early adulthood). The fat mass was used as our primary outcome hypothesis. Lean mass was used as a sensitivity analysis or comparison outcome to assess whether the associations with fat mass were distinct from those shown with lean mass.
Statistical analyses
As we have indicated, earlier analyses using this cohort had shown associations between the fathers’ onset of regular smoking pre-puberty and increased fat mass in their offspring. Here we extend this analysis to demonstrate the associations between age at commencement of smoking and the outcomes to their grandchildren and great grandchildren. Given the findings with the fathers’ prepubertal onset of regular smoking, we examined possible associations with the pre-puberty onset of smoking of the two grandfathers. Because there is evidence that puberty started at later ages in the first half of the twentieth century21, we took < 13 years to denote the pre-puberty ages. Unfortunately, the numbers were too small for valid analysis for the paternal great-grandfathers PGFF and PGMF.
In general, we found extremely few grandmothers and great-grandmothers who had begun smoking prepuberty—numbers were too small for valid analysis. The numbers of reports of the age at onset of smoking of grandfathers and the maternal great-grandfathers pre-puberty were greater and were deemed sufficient for unadjusted analyses. The smoking information available only included whether the ancestor had started smoking in childhood (< 17) but did not include later onset. The analyses have therefore concentrated on comparing the families where the ancestor had started smoking aged < 13 (pre-puberty) with controls whose ancestors had started smoking in adolescence (13–16 inclusive); the ancestors of this generation who had either not smoked at all or who had started smoking after the age of 16 were omitted.
Because of the very small numbers in the index group, and lack of power, it was decided to consider 1-tailed P values of ≤ 0.10 since the study hypothesis concerned an increase (not a decrease) in fat mass. We had no prior hypotheses in regard to lean mass, so the tests were 2-tailed.
Initial analyses determined the unadjusted associations between each of the two grandfathers (F1s) and those of the two maternal great-grandfathers (F0s) where the numbers smoking pre-puberty were sufficient. The F3 outcomes were calculated separately using multiple regression analyses for (i) all children, (ii) males and (iii) females. In each instance the mean difference (MD) for both the fat and the lean mass of the target individuals (i.e., those whose ancestor started smoking pre-puberty) were compared with those who started smoking later in childhood using 95% confidence intervals. Presence of a significant interaction between the sexes was assumed when both the MD of the males was outside of the 95% confidence interval of the females and vice versa.
The numbers of individual male ancestors who had started to smoke in childhood at < 13 years and for whom data on age at starting to smoke regularly before age 17 were available altogether are shown in Table 1. The numbers smoking prepuberty were small, especially when stratified by sex. The grandparents had greater numbers for each sex, with maternal grandfathers having almost twice the totals of paternal grandfathers. Only two of the great-grandparents had sufficient numbers for analysis, both were on the maternal side. Consequently, the study was more likely to demonstrate evidence of association of prepubertal smoking in the maternal rather than the paternal line.
A further set of stratified analyses were carried out to determine whether associations shown with fat mass were the consequence of inherited likelihood of starting to smoke pre-puberty by omitting the great-grandchildren whose grandparents had started smoking pre-puberty. The lines of heredity shown in Fig. 2 show that, if our results are repeated, there is no possibility of a direct effect of a marker on the X chromosome of the paternal grandfather or great-grandfather, but that epigenetic markers on the autosomes are feasible.