Statistics
Statistical tests are two-sided Welch’s t-tests unless otherwise specified. Enrichment analyses were performed using the phyper function for hypergeometric tests in R. Multiple testing correction was performed based on the Benjamini–Hochberg method. Biomarker analysis was performed using GDSCTools as described below. Box plots depict the median value as the centre bar, first and third quartiles as box boundaries, and whiskers extending to either first quartile minus 1.5 × the inter-quartile range (lower boundary), or third quartile plus 1.5 × the inter-quartile range (upper boundary). Points beyond this region are individually plotted.
Cell lines
Cell lines were acquired from commercial cell banks. All cells were grown in RPMI medium (supplemented with 10% FBS, 1% penicillin/streptomycin, 1% glucose, 1 mM sodium pyruvate) or DMEM/F12 medium (supplemented with 10% FBS, 1% penicillin/streptomycin) (Supplementary Table 2) at 37 °C in a humidified atmosphere at 5% CO 2 . To prevent cross-contamination or misidentification, all cell lines were profiled using a panel of 94 single nucleotide polymorphisms (Fluidigm, 96.96 Dynamic Array IFC). Short tandem repeat (STR) analysis was also performed, and cell line profiles were matched to those generated by the cell line repository. All cell lines are routinely tested for mycoplasma and are negative for mycoplasma. Further information on the cell lines used in this study, including their source and molecular profiling datasets can be found in Supplementary Table 2 and in the Cell Model Passports database30 (https://cellmodelpassports.sanger.ac.uk).
Compounds
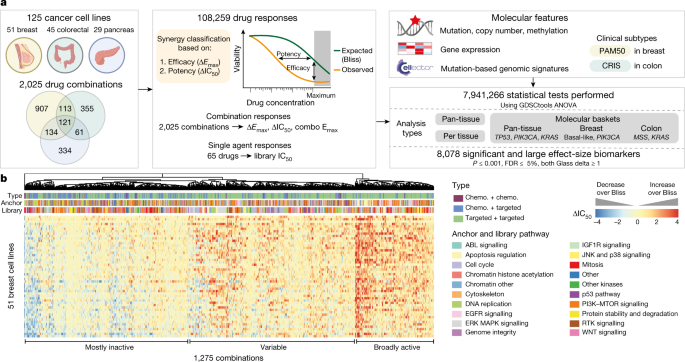
Compounds were sourced from commercial vendors (Supplementary Table 1). DMSO-solubilized compounds were stored at room temperature in low humidity (<12% relative humidity), low oxygen (<2.5%) environment using storage pods (Roylan Developments). Water-solubilized compounds were maintained at 4 °C. For 8 compounds their identities and purity were confirmed by UHPLC-MS. Identity was confirmed by mass spectrometry (6550 iFunnel Q-TOF LC/MS, Agilent Technologies) using electrospray ionization in positive and/or negative modes. Anchor and library concentrations were drug- and tissue-specific and determined using a two-step process. First, drug concentrations were selected based on primary literature, in vitro data of minimum concentrations inhibiting relevant target activity and viability31, clinical data indicating achievable human plasma concentrations, or where known concentrations that induce sensitivity in a biomarker positive cell line. Additionally, a pilot screen, testing a 1,000-fold concentration range of each drug in 9–13 cell lines per tissue (breast: 13, colon: 9, pancreas: 12), was performed and concentrations optimized to give a range of sensitivities across the cell lines. Anchor drugs were screened at two fixed concentrations with a 2-, 4- or 10-fold difference between them to give moderate activity (50–90% viability) across the cell lines within each cancer type. Screening concentrations (Supplementary Table 1) typically did not exceed 10 μM and were in the range of human plasma exposures achievable in patients32. Library drugs were screened at seven concentrations spanning a 1,000-fold range with a non-equidistant log 2 design of four 4-fold steps followed by two 2-fold dilution steps starting at the lowest used concentration. The use of this design was based on the observation that higher concentrations were most informative and would benefit from denser profiling. As an alternative to μM concentration ranges, drug concentrations and IC 50 values can be visualized on a standardized log 2 scale, with 9 being equivalent to the highest screened concentration. Each screening plate contained five replicates of the anchor alone (high and low concentrations) and four replicates of the library alone (full dose response). A single replicate of the combination dose response was performed in the primary screen.
Screening
Cells were transferred into 1,536-well plates in 7.5 μl of their respective growth medium using XRD384 (FluidX) dispensers. The seeding density was optimised prior to screening to ensure that each cell line was in the exponential growth phase at the end of the assay. For this, six seeding densities with a two-fold dilution step were each dispensed into 224 wells of a single 1,536-well assay plate (XRD384 (FluidX) dispenser) and cells were incubated for 96 h. Cell number was quantified using CellTiter-Glo 2.0 (Promega). The maximum density tested varied based on cell type, typically 5,000 cells per well for suspension cells and 1,250 cells per well for adherent cells (Supplementary Table 1). Assay plates were incubated at 37 °C in a humidified atmosphere at 5% CO 2 for 24 h then dosed with the test compounds using an Echo555 (Labcyte). Final DMSO concentration was typically 0.2%. Following dosing with compounds assay plates were incubated, and the drug treatment duration was 72 h. To monitor cell growth over the duration of drug treatment, a parallel undrugged control plate was assayed at the time of drug treatment and referred to as a ‘day = 1’ plate. This was repeated each time that a cell line was screened. To measure cell viability, 2.5 μl of CellTiter-Glo 2.0 (Promega) was added to each well and incubated at room temperature for 10 min; quantification of luminescence was performed using a Paradigm (Molecular Devices) plate reader.
Assay plate quality control
All screening plates contained negative control wells (untreated wells, n = 6; DMSO-treated wells, n = 126) and positive control wells (blanks—that is, medium-only wells, n = 28; staurosporine-treated wells, n = 20; and MG-132 treated wells, n = 20) distributed across the plate. We used these positive and negative control wells to test whether the plates meet defined quality control criteria. A maximum threshold of 0.18 was applied to the coefficient of variation (CV) of the DMSO-treated negative controls (CV = σ N /μ N , where σ N is the s.d. of the negative control and μ N is the mean of the negative control). Using the DMSO-treated negative control (NC1) and the two positive controls (PC1 and PC2), we determined Z-factors (also known as Z′; Z-factor = 1 – 3 × (σ P + σ N ) / (|μ P − μ N |), where σ N and σ P are the s.d. of the negative and positive controls, respectively, and μ N and μ P are the mean of the negative and positive controls, respectively). The Z-factors were calculated for all plates that indicate sensitivity of the cell lines to the positive control (ratio of NC1:PC ≥4). In case a cell line is insensitive to both positive control drugs, the Z-factors were calculated based on blank wells instead. Z-factors were required to exceed a minimum threshold of 0.3 for individual plates and a mean of 0.4 across all plates within a screening set. Where a cell line was sensitive to both positive controls, it had to pass Z-factor thresholds for both positive controls. Plates that did not meet these requirements were excluded from the study. Overall, 3,106 (>70%) of 1,536-well microtitre screening plates passed coefficient of variation and Z-factor thresholds. Wherever possible, failed plates were repeated, leading to dataset completeness of more than 96% for all three tissues (breast: 96.5%, colon: 99.8% and pancreas: 99%).
Curve fitting
For each plate, the raw fluorescent intensity values were normalised to a relative viability scale (ranging from 0 to 1) using the blank (B) and negative control (NC) values (viability = (Fluorescence of treated cells − B)/(NC − B)). Anchor viability was determined from the mean across the five replicate wells screened on each plate. All library drug dose responses were fitted as a two-parameter sigmoid function33. The dose–response curves for the combinations were fitted similarly, butwith two notable differences: (1) the cell line parameters were obtained from the library drug fits; (2) the maximum viability was capped at the anchor viability (rather than from 0 to 1). We use the 50% (inflection) point of the sigmoidal curve between zero and the anchor viability for both the expected Bliss and the observed combination. We extended the model to nest each replicate within the drug or cell line to obtain stable estimates from the replicate experiments. To assess the quality of the fits, we computed the root mean square error (RMSE) and excluded curves with RMSE > 0.2 (equalling 1.5% of measurements). The E max and the IC 50 are based on the fitted curves. E max is reported at the highest tested concentration for the drug.
Classifying synergy
To detect synergy we compared observed combination responses to expected combination responses. For the latter, we used Bliss independence9 of the response to the anchor and the library drug alone. Conceptually, every point on the Bliss dose response curve is defined as the product between the anchor viability and the corresponding point on the library dose response curve. Shifts in potency (∆IC 50 ) and in efficacy (∆E max ) were calculated as the difference between the observed combination response and expected Bliss (∆IC 50 = Bliss IC 50 − combination IC 50 , and ∆E max = Bliss E max − combination E max ). ∆IC 50 is reported on a log 2 scale.
A given measurement was synergistic if the combination IC 50 was less than twice the highest screened library concentration and either the ∆IC 50 or the ∆E max was above a specific threshold: ∆IC 50 ≥ 3 (23 is equivalent to an 8-fold shift in IC 50 ) or the ∆E max ≥ 0.2 (20% shift in viability). Replicate measurements of ‘anchor-library–cell line’ tuples were summarized as synergistic if half or more of the replicate measurements showed synergy. To summarize both anchor concentrations, we considered a ‘combination–cell line’ pair as synergistic if synergy was observed at either anchor concentration.
Reproducibility
To assess the reproducibility within a screen, we generated 2–18 biological replicates for 4–5 cell lines per tissue (breast: 5 (AU565, BT-474, CAL-85-1, HCC1937, MFM-223); colon: 4 (HCT-15, HT-29, SK-CO-1, SW620); pancreas: 5 (KP-1N, KP-4, MZ1-PC, PA-TU-8988T, SUIT-2)). Single-agent and combination responses were averaged across technical replicates (typically 3 per biological replicate) and correlated (Pearson correlation coefficient; minimum of 322 biological replicate pairs per ‘metric-tissue’ pair).
To assess the reproducibility of the screen, we rescreened a subset of combinations in each tissue (breast: 51 combinations in 34 cell lines; colon: 45 combinations in 37 cell lines; pancreas: 59 combinations in 29 cell lines; Supplementary Table 2). Drug combination responses were averaged across replicates within a screen and key metrics of single-agent and combination response were correlated between the two screens (Pearson correlation coefficient). To determine the quality of synergy calls, the original screen was considered as ground truth and numbers of true positive (TP), false positive (FP), true negative (TN) and false negative (FN) synergistic combination–cell line pairs were calculated. These were used to calculate F-score (F-score = TP/(TP + 0.5 × (FP + FN))), recall (recall = TP/(TP + FN)), and precision (precision = TP/(TP + FP)) per tissue. To investigate the strength of effects of ΔE max and ΔIC 50 of FP and FN measurements, the distance to ΔE max and ΔIC 50 synergy thresholds was calculated for each ‘anchor concentration-library–cell line’ tuple based on combination responses averaged across replicates (n = 9,570 tuples).
Biomarker analysis
Matrices of single-agent (library IC 50 ) and combination response (combination E max , ∆IC 50 , ∆E max ) metrics were used as input for GDSCTools ANOVAs15. To obtain a single combination E max , ∆IC 50 and ∆E max value per cell line–combination pair, responses were averaged across replicates for each anchor concentration-library–cell line tuple and the combination metrics were compared for the two anchor concentrations: the larger of the two ∆IC 50 and ∆E max values and the smaller of the two combination E max values were used for biomarker discovery in order to capture the largest effects of the combination. A range of binary feature files were used, including multi-omics binary event matrices (MOBEMs) composed of genes known to be mutated, amplified or homozygously deleted in human cancers5 (number of features = 1,073), CELLector signatures34 (n = 227 for breast, n = 261 for colon), RNA-seq gene expression35 (n = 1,184; original dataset accession number E-MTAB-3983), CRIS36 and PAM5037,38 classifications. Gene expression was limited to a curated panel of genes composed of targets of the drugs used, additional members of the BCL2 family and apoptosis-associated genes39, genes annotated as clinically relevant for cancer40, and genes whose mutations were listed as features in the MOBEMs5,40 and CELLector5,34,40 feature files. Continuous values of gene expression were binarized by z-scoring each variable across the subset of cell lines used for the molecular context tested, and substituting a z-score ≥ 2 for a binary value representing that feature being elevated (that is, ‘Gene_up’), and a z-score ≤ −2 for a binary feature representing that feature being decreased (that is, ‘Gene_down’). Overall significance thresholds were P ≤ 0.001 and FDR ≤ 5%.
Network overlays
An interactome of binary, undirected interactions was built in the iGraph R package (https://cran.r-project.org/web/packages/igraph/citation.html) using the Reactome18 human interactions file (accessed April 2021), and all human interactions reported in IntAct12 (accessed July 2021). All non-protein nodes and duplicated interactions were removed, resulting in a non-directed network of 5,556 Uniprot protein nodes and 25,731 edges for the Reactome interactome. For the IntAct interactome, an evidence filter of 0.5 was applied, and all non-protein nodes and duplicate interactions were removed, resulting in a non-directed network of 14,431 protein nodes and 110,118 edges. Drug targets and biomarkers features were manually mapped to their Uniprot proteome identifiers (UPID), with overall 57 out of 66 (86.3%) drug target profiles being mapped to one or more UPIDs, and 633 out of 1,501 (42.2%) biomarker features being mapped for one or more UPIDs. UPID mapping was not possible for chemotherapeutics, PAM50, CRIS, and not done for methylation sites not associated with a cancer driver gene. A distance matrix between all nodes was calculated using iGraph: infinite values were reported for nodes that did not exist in the same network. When calculating the shortest distance between drug targets or drug targets and biomarkers, distances were calculated for all target-target or target-biomarker pairs and the smallest distance was reported. For example, for a drug with two targets combined with a drug with three targets, the shortest of six target-target distances would be reported. To simulate false positive biomarker associations, the biomarker features used in the genuine distance plot were randomly shuffled without replacement, before re-calculating the shortest distance between the new, ‘false’ biomarker and drug targets.
Clinical trials
Clinical trials data were extracted from the API at https://clinicaltrials.gov/ (accessed March 2021) using an R script and searches in the format ‘drug1 + drug2 + cancer + tissue’. Obtained lists of trials were manually curated to ensure that drugs were exact matches and to remove trials using radiotherapy alongside drug combination treatment. Searches were limited to 81 combinations with ≥ 25% synergy in populations of clinical need.
Specificity of CHEK inhibition
To test CHEK specificity we seeded SW620, SW837, SNU-81 or LS-1034 cells in 96-well plates (770–2,750 cells per well) and treated them with camptothecin (anchor, 0.025 μM) in combination with six CHEK inhibitors (libraries, all dosed at 1 μM highest used concentration unless indicated): AZD7762 (CHEK1, CHEK2), prexasertib (CHEK1, CHEK2), MK-8776 (CHEK1, CHEK2, CDK2), SAR-020106 (CHEK1), rabusertib (CHEK1) and CCT241533 (CHEK2; 2 μM). After 96 h of drug treatment viability was measured with CellTiter-Glo 2.0 (CTG; Promega). Drug response curves were fitted as described above.
For siRNA experiments, SW837 and SNU-81 cells (8,000 and 16,000 cells per well, respectively) were reverse transfected with siRNAs of a non-targeting pool as negative control (siNT; Dharmacon, D-001810-10-05), polo-like kinase 1 (PLK1) pool as positive control (Dharmacon, L-003290-00-0010), CHEK1 pool (Dharmacon, L-003255-00-0005) or CHEK1 individualsiRNAs (LQ-003255-00-0005), and CHEK2 pool (Dharmacon, L-003256-00-0005) using lipofectamine RNAiMax (Thermofisher). After 30h, 0.025 μM or a dose range of 0.001–9 μM SN-38 or DMSO were added and viability was measured 72 h later with CTG. Signal was normalised to siNT+DMSO controls. Statistical significance between conditions was tested using a two-sided Welch’s t-test.
Real time cell death and caspase-3/7 activity
Cells were seeded in 96-well plates (typically 5,000–16,000 cells per well). After 24 h drugs (0.125 μM staurosporine (positive control), 0.025 μM SN-38, 0.75 μM rabusertib, 0.75 μM CCT241533) or DMSO and real-time fluorescent reagents for detection of cell death (CellTox Green; 1:1,000; Promega) or caspase-3/7 activity (IncuCyte Caspase-3/7 Red; 1:1,000; Essen Bioscience) were added. Pictures were recorded every 2 h for 96 h using an Incucyte (Essen Bioscience). Recorded fluorescent signals were measured as mean intensity per cell area and normalised to time 0 h.
Colony formation
Cells were seeded in 6-well plates at 50,000 cells per well. Drugs (0.1 nM SN-38, 0.5 μM rabusertib, 0.5 μM CCT241533) or DMSO were added on day 1 and were refreshed through medium change on day 8. 14 days after drug treatment started the cells were fixed in 4% paraformaldehyde (Sigma-Aldrich) in PBS for 10 min at room temperature, and then stained with Giemsa (10%; Sigma-Aldrich) for at least 30 min at room temperature.
Western blot
SW837 (1 million) or SNU-81 (1.5 million) cells were seeded in 10 cm dishes and treated with drugs (0.025 nM SN-38, 1.5 μM rabusertib, 1.5 μM CCT241533, 2 μM MG-132 (positive control)) or DMSO the day after. After 72 h, live and dead cells were collected and lysed in RIPA buffer (Sigma-Aldrich) supplemented with 1 mM DTT (Cayman Chemicals) and protease and phosphatase inhibitors (Roche). Total protein content was determined using Bradford reagent (ThermoFisher) and around 20 μg of lysate were loaded onto a 4–12% Bis-Tris gel (Invitrogen) for SDS–PAGE followed by protein transfer from the gel onto a PVDF membrane. Membranes were blocked in 5% milk (in TBST) and incubated overnight with the appropriate antibodies. Blots were washed in TBST and incubated with secondary antibody for 1 h at room temperature. Blots were washed in TBST before the signal was enhanced with Super Signal Dura and visualised. The following primary antibodies were used for immunoblot analysis: anti-PARP (Cell Signaling Technologies, 9542, 1:1,000; rabbit), and anti-β-tubulin (Sigma-Aldrich, T4026, 1:5,000; mouse) as loading control.
For experiments with knockdown of CHEK1 and CHEK2, SW837 or SNU-81 cells were reverse transfected with siNT, siCHEK1 or siCHEK2 as described above. Cells were collected 72 h after transfection and lysed in RIPA buffer (Sigma-Aldrich, R0278) and protein concentrations were determined using the BCA assay (Novagen, 71285-3) as per manufacturer’s instructions. SDS–PAGE and western blots were conducted as described above and the following primary antibodies were used for immunoblot analysis: anti-CHEK1 (Santa Cruz Biotechnology, sc-8408, 1:200; mouse), anti-CHEK2 (Cell Signaling Technologies, D9C6, 1:1,000; rabbit), and anti-β-actin (Abcam, ab6276, 1:5,000; mouse) as a loading control. Anti-Mouse IgG (GE Healthcare, NA931) and anti-rabbit (GE Healthcare, NA934; 1:2,000) HRP-linked secondary antibodies were used as secondary antibodies. PageRuler Plus Prestained Protein Ladder, 10–250 kDa (ThermoFisher, 26620) was used as a molecular weight marker.
In vivo tumour xenograft studies
A total of 4.5 × 106 LS-1034 cells, 5 × 106 SW837 cells or 2.5 × 106 SNU-81 cells in 30% Matrigel were injected subcutaneously into the right flank of male and female 6-week-old NOD/SCID mice. Once tumours reached an average volume of approximately 300–400 mm3, mice were randomized into treatment arms, with n = 12 (irinotecan and irinotecan + rabusertib) or n = 6 (vehicle and rabusertib) per group. Rabusertib was administered orally, 200 mg kg−1 daily (vehicle: 16.66% Captisol; CyDex, in 25 mM phosphate buffer, pH 4); irinotecan was administered intraperitoneally, 25 mg kg−1 twice a week (vehicle: physiological saline). Tumour size was evaluated once weekly by calliper measurements, and the approximate volume of the mass was calculated using the formula (4π/3) × (d/2)2 × (D/2), where d is the minor tumour axis and D is the major tumour axis. Results were considered interpretable when a minimum of 4 mice per treatment group reached the prespecified endpoints (at least 3 weeks on therapy or development of tumours with average volumes larger than 2,000 mm3 within each treatment group in trials aimed to assess drug efficacy; at least 3 weeks after treatment cessation or development of individual tumours with volumes larger than 750 mm3 in survival experiments aimed to assess tumour control by therapy). A major tumour axis of 20 mm is the endpoint permitted by the Italian Ministry of Health in authorization no. 806/2016-PR, in accordance with national guidelines and regulations. This endpoint was not exceeded in any experiment. Operators were blinded during measurements. In vivo procedures and related biobanking data were managed using the Laboratory Assistant Suite41. All animal procedures were approved by the Institutional Animal Care and Use Committee of the Candiolo Cancer Institute and by the Italian Ministry of Health.
Statistical significance for tumour volume changes during treatment was calculated using a two-way ANOVA. For endpoint comparisons, statistical analysis was performed by two-tailed unpaired Welch’s t-test. Statistical analyses in the survival experiments were performed by log-rank (Mantel–Cox) test. For all tests, the level of statistical significance was set at P < 0.05. Graphs were generated and statistical analyses were performed using the GraphPad Prism (v9.0) statistical package.
Immunohistochemistry
Morphometric quantitation of Ki67, active caspase-3, and phospho-H2AX immunoreactivity was performed in xenografts from mice treated with vehicle (until tumours reached an average volume of 1,500 mm3) or the indicated compounds (after 72 h). Tumours (n = 1–3 for each treatment arm) were explanted and subjected to histological quality check and immunohistochemical analysis with the following antibodies: mouse anti-Ki-67(MIB-1) (Dako, GA626, 1:100), rabbit anti-cleaved caspase-3 (Asp175) (Cell Signaling, 9661, 1:200) and rabbit anti-phospho-histone H2AX (Ser139) (20E3) (Cell Signaling, 9718, 1:400). After incubation with secondary antibodies, immunoreactivities were revealed by DAB chromogen (Dako). Images were captured with the Leica LAS EZ software using a Leica DM LB microscope. Morphometric quantitation was performed by ImageJ software using spectral image segmentation. Software outputs were manually verified by visual inspection of digital images. Each dot represents the value measured in one optical field (40× for Ki67 and phospho-H2AX; 20× for active caspase-3), with 2–10 optical fields (Ki67 and phospho-H2AX) and 3–5 optical fields (active caspase-3) per tumour depending on the extent of section area (n = 12–30 for Ki67 and phospho-H2AX; n = 8–15 for active caspase-3). The plots show mean ± s.d. Statistical analysis by two-tailed unpaired Welch’s t-test.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this paper.