Purine nucleobases and related molecules in the Murchison extracts
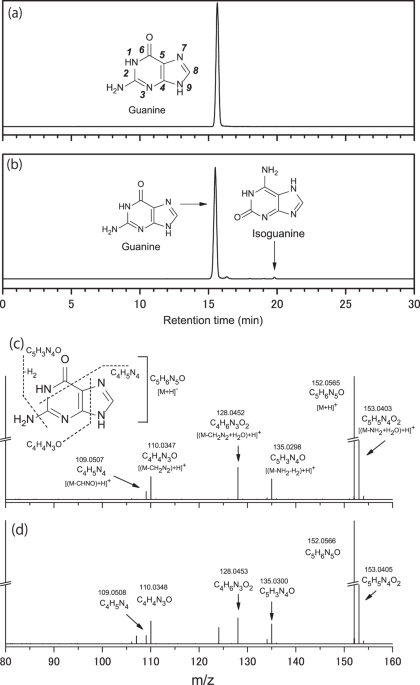
We detected several purine nucleobases and their analogs in both Murchison meteorite extracts (Fig. 1, Supplementary Fig. S2, and Table 1) based on their chromatographic retention time, accurate mass measurements of their parent masses, and mass fragmentation patterns in the MS/MS measurements (Fig. 1b). Guanine is typically the most abundant purine nucleobase (hereafter referred to as purine molecules, unless otherwise denoted) in all meteorites (except for Tagish Lake), followed by adenine, xanthine, and hypoxanthine as the second most abundant group, with purine, isoguanine, and 2,6-diaminopurine as the least abundant group. The total concentrations of purine molecules were 152 and 11 ppb in the aqueous extracts of two Murchison meteorite specimens (one obtained from the University of Chicago and the other from a meteorite trading company. Hereafter, each specimen is denoted as Murchison #1 and Murchison #2, respectively). Although the concentrations in the Murchison #1 extract and those reported by Callahan et al.19 are similar, those in the Murchison #2 extract were generally one order of magnitude lower. Because the analytical conditions used for the two Murchison specimens in the present study were essentially identical, we can infer that the observed difference was due to sample heterogeneity within the Murchison meteorite (e.g., meteoritic amino acid profiles of Murchison)8,27. Significant heterogeneity in amino acid abundance was previously reported in different lithologies of the Tagish Lake meteorite, where total amino acid concentrations ranged from 40 ppb in the most aqueously altered samples up to 5400 ppb in the least altered lithologies28. The weighted average of the concentrations of purine molecules in the two Murchison specimens is shown in Table 1 and has a similar order of magnitude as that reported by Callahan et al.19. Note that there is a striking difference between the extraction method used in the current study and that used by Callahan et al.19, who extracted meteorites in 95% formic acid at 100 °C for over 24 h. If such severe extraction conditions are more suitable for the extraction of nucleobases from meteorites, it is likely that the actual concentrations of nucleobases in our meteorite samples are higher than those reported here. Nevertheless, in the present study, we used a less harsh extraction method to prevent the undesirable decomposition of fragile molecules such as sugars6 and hexamethylenetetramine26 present in the meteorite specimens. Consequently, the weighted average concentrations in our Murchison extracts were lower than those reported previously. We concluded that the detection of purine (C 5 H 4 N 4 ) in the meteorite extracts strongly supported the indigenous nature of the detected purine molecules in the Murchison meteorites19.
Fig. 1: Detection of purine nucleobases. Mass chromatograms at m/z = 152.0567 within a 3 ppm exact mass window at the monoisotopic mass for a the guanine (0.21 ng) standard reagent (STD) and b Murchison #1 extract were measured using an InertSustain PFP column. Additionally, isoguanine, a structural isomer of guanine, was detected in the Murchison #1 extract. Mass fragmentation patterns of c guanine STD and d guanine were detected in the Murchison #1 extract measured by MS/MS experiments. Note: “exact mass”, representing a theoretical value obtained by calculation, is different from “accurate mass”, which is a measured value with a high-resolution mass spectrometer. Bold italic numbers in panel a represent the numbering of atoms in the chemical structures of purine nucleobases. Full size image
Table 1 Variations in the concentration (in ppb) of purine nucleobases and their analogs in the meteorite extracts and Murchison soil. Full size table
Pyrimidine nucleobases in the Murchison extracts
We detected a wide variety of pyrimidine nucleobases and their structural isomers from both Murchison extracts (Fig. 2, Supplementary Figs. 3, 4, and Table 2), most of which had not been previously detected in meteorites. Uracil is the only pyrimidine nucleobase previously quantified in the Murchison meteorite (30 ppb in an H 2 O extract)29. Although the abundance was not quantified, uracil was also detected in the formic acid extracts of the Murchison meteorite, and the stable carbon isotopic composition (δ13C) was measured to be +44.5‰ (vs. VPDB standard)18. The uracil concentrations in the Murchison #1 and #2 meteorites were 15 and 1 ppb, respectively, showing that uracil is distributed heterogeneously in the Murchison meteorite. The weighted average of the uracil concentration (7 ppb) was approximately four times lower than that reported in the previous study29, likely due to Murchison sample heterogeneity. In addition, the uracil concentration in the previous study could have been overestimated because of the co-elution of other molecules, such as carboxylic acids. As shown in Fig. 2a, in addition to uracil, we detected the structural isomers 2-imidazolecarboxylic acid and 4-imidazolecarboxylic acid in the same extract, and their concentrations exceeded that of uracil (Table 2). If the peak separation between uracil and its isomers is inadequate, as in the case shown in Supplementary Fig. 5, the uracil concentration would be overestimated. Hence, further confirmation using an appropriate LC condition (Fig. 2a) and MS/MS measurements (Fig. 2b and Supplementary Fig. 4) are required to identify a specific molecule in a mixture of complex molecules containing structural isomers.
Fig. 2: Detection of pyrimidine nucleobases. Mass chromatograms at m/z = 113.0346 within a 3 ppm exact mass window at each monoisotopic mass for a the uracil (0.46 ng) standard reagent (STD) and b Murchison #1 extract measured using a Hypercarb column. In addition to uracil, 4-imidazolecarboxylic acid and 2-imidazolecarboxylic acid were detected as the dominant structural isomers in the Murchison #1 extract. Mass fragmentation patterns of c uracil STD and d uracil were detected in the Murchison #1 extract measured by MS/MS experiments. Mass peaks with a star (*) represent background signals in the fragmentation spectra present in both the meteorite and reference STD spectra. Bold italic numbers in panel a represent the numbering of atoms in the chemical structures of pyrimidine nucleobases. Full size image
Table 2 Variations in the concentration (in ppb) of pyrimidine nucleobases and their related analogs in meteorite extracts and the Murchison soil. Full size table
Other pyrimidine nucleobases, such as cytosine and thymine, as well as their analogs containing a pyrimidine ring, were identified by their chromatographic retention times, accurate mass measurements of their parent masses, and mass fragmentation patterns in the MS/MS measurements (Supplementary Table 1), with concentrations ranging from 0.02 to 6 ppb (Table 2). Methylated pyrimidine nucleobases (except thymine, a methylated form of uracil), such as 5-methylcytosine, 1-methyluracil, and 6-methyluracil, are less common in biological systems than major bases such as cytosine and uracil30,31,32. The observed similar concentrations of methylated and non-methylated pyrimidine nucleobases strongly suggest that they are abiological and hence extraterrestrial in origin. Though the presence of the relatively unstable cytosine33 is unexpected and would typically be attributed to contamination, their presence in abundances similar to isomers and methylated analogs reinforces the abiological origin of cytosine. The reason for the preservation of cytosine is unclear, perhaps it was produced at a steady state in the meteorite parent bodies and protected from deamination due to rapid drying and/or low temperatures once the hydrothermal phase on the asteroid had stopped. In addition, some coexisting species on the asteroid could have a role for the preservation of cytosine, which will be investigated in our future study.
The total concentrations of the detected pyrimidine nucleobases and their analogs (hereafter referred to as pyrimidine molecules, unless otherwise denoted) were 37 and 15 ppb in Murchison #1 and Murchison #2, respectively, and the weighted average was 24 ppb, which is ~0.1% of the total amino acid concentration in the Murchison meteorite3. The much lower concentrations of pyrimidine and purine molecules compared to meteoritic amino acids suggest that nucleobases are less suitable for formation in extraterrestrial environments, including meteorite parent bodies. Pyrimidine (C 4 H 4 N 2 ) was not identified in the two Murchison extracts, which is consistent with organic residues in UV-processed interstellar ice analogs25. The absence of pyrimidine in meteorite extracts suggests that the addition of side chains such as methyl (-CH 3 ) and amino (-NH 2 ) groups to pyrimidine may not be the dominant pathway to the formation of the detected pyrimidine nucleobases.
Assessment of terrestrial contamination of nucleobases in the Murchison extract
The analysis of extracts from the procedural blank that consisted of prebaked quartz did not yield any detectable nucleobases, indicating no terrestrial nucleobase contamination during the analytical procedures. However, we detected pyrimidine and purine nucleobases from the Murchison soil extract at concentrations ranging from 1 to 141 ppb (Tables 1 and 2), which were generally higher than those in the Murchison extracts (Tables 1 and 2). Callahan et al.19 also detected purine and pyrimidine nucleobases in the Murchison soil formic acid extract at concentrations ranging from 7 to 1380 ppb, with cytosine being the most abundant. In contrast, except for DNA/RNA nucleobases, the Murchison soil showed less molecular diversity than the formic acid extract of the Murchison meteorite. Based on significant differences in the distributions of nucleobases and their analogs between the Murchison meteorite and soil extracts, including purine, 2,6-diaminopurine, and 6,8-diaminopurine, which were detected in the meteorite extract but not in the soil, they concluded that purines found in the Murchison meteorite are extraterrestrial in origin. We used two Murchison meteorite specimens: a 2 g specimen from the University of Chicago (Murchison #1) and a 2.7 g specimen from a meteorite trading company (Murchison #2). If terrestrial contamination dominates the meteoritic nucleobase distribution in the present study, it indicates that the meteorite size and nucleobase concentration are positively correlated. However, the actual distributions were opposite, with the concentrations of nucleobases in Murchison #1 generally being much higher than those in Murchison #2 (Tables 1 and 2); this strongly suggests that the nucleobases in the soil are not inherited by the meteorite and hence the detected nucleobases are all extraterrestrial in origin. In addition, other purine and pyrimidine molecules, such as isoguanine, 5-methylisocytosine, and purine, were not detected in the soil extracts, supporting the assumption described above. Note that alanine extracted from Murchison #1 was racemic34, which strongly suggests that the detected alanine is extraterrestrial in origin with no evidence of significant biological contamination of the meteorite sample. This would also be the case for nucleobases. Moreover, in general, the higher concentration of dihydrouracil compared to nucleobases cannot be explained by terrestrial biology35. Hence, we conclude that the observed chemical diversity of the purine and pyrimidine molecules and their isomers are indigenous and suggests an extraterrestrial origin, consistent with the detection of racemic amino acids and primordial sugars from the same extracts6,7.
Purine and pyrimidine molecules and their isomers in the Tagish Lake and Murray extracts
Various purine and pyrimidine molecules were also identified in the Tagish Lake and Murray extracts (Tables 1 and 2). Guanine was the most abundant in the Murray extract (25 ppb), as is the case for the two Murchison extracts, whereas, for the Tagish Lake extract, adenine was the most abundant (6 ppb) purine molecule and 6-methyluracil (17 ppb) is the most abundant pyrimidine molecule. The Tagish Lake extract generally shows less diversity of purine and pyrimidine molecules compared with the Murchison and Murray extracts, and their absolute concentrations are generally lower than those of the other meteorite extracts. The observed trend in molecular abundance and diversity is consistent with that observed for amino acids in these three meteorites28, suggesting that the Tagish Lake and the other two meteorites may have experienced distinct processes on their parent bodies. The total concentrations of detected purine molecules in the Tagish Lake and Murray extracts were 9 and 33 ppb, respectively, and those of pyrimidine molecules were 37 and 13 ppb, respectively. Note that there is a prominent difference in the (total pyrimidine molecules)/(total purine molecules) ratio between the Tagish Lake and the other two meteorites: for the former, pyrimidine molecules dominate over purine molecules, whereas the opposite is true for the latter. This observed difference may reflect different formation pathways, as discussed later.
Important nitrogen heterocyclic molecules in the meteorite extracts
Imidazole is an interesting molecule in terms of a primitive catalyst for molecular syntheses36, which might be a potential precursor of purine formation37. Due to its potential importance, it has been a target of multiple astrochemical25,37 and astronomical studies38. Here, we identified not only imidazole itself but also its related analogs, some of which are the structural isomers of pyrimidine nucleobases such as cytosine, uracil, and thymine at typically higher concentrations than the corresponding pyrimidine bases (Fig. 2 and Supplementary Fig. 3). For example, 4-imidazolecarboxylic acid and 1-methyl-1H-imidazole-5-carboxylic acid, which are structural isomers of uracil and thymine, respectively, are 1–2 orders of magnitude more abundant than the corresponding pyrimidine nucleobases (Table 2). This strongly suggests the presence of preferential routes for the formation of imidazole-containing molecules over pyrimidine-containing molecules in extraterrestrial environments. The reactivity of imidazole and the contribution of electron-conjugated systems are assumed to induce progressive cyclization and variation in purine derivatives. Indeed, imidazole and its alkylated homologs are much more abundant than pyrimidine nucleobases (Table 3), supporting our hypothesis. We estimated that the progression of the N-heterocyclic reaction would correlate with the thermal history of the parent body.
Table 3 Variations in the concentrations (in ppb) of imidazole and its alkylated analogs. Full size table
We also confirmed the presence of alkylated homologs of imidazole using the same procedure described above (Supplementary Fig. 6). Larger alkylated imidazole homologs were observed in the mass chromatograms using a retention index of systematic methylene (-CH 2 -) elongation (Supplementary Fig. 7). These results are qualitatively consistent with the tentative detection of alkylated imidazole homologs (i.e., imidazole with <C 15 ) in the Murchison meteorite9. Given that the observed peaks on each mass chromatogram are solely derived from alkylated imidazole homologs with identical ionization efficiencies, the total concentrations of alkylated imidazole homologs could be as high as 41,300, 500, and 21,200 ppb in the Murchison, Tagish Lake, and Murray extracts, respectively. This implies that alkylated imidazole homologs are one of the most abundant classes of solvent-extractable organic compounds in these three carbonaceous meteorites. Naraoka et al.9 proposed a possible pathway for the formation of alkylimidazoles via the Radziszewski reaction, which uses aldehydes and ammonia as the initial reactants. Since aldehydes and ammonia might be formed by the hydrothermal decomposition of HMT in meteorite parent bodies26, the formation of alkylimidazoles could be enhanced under the same environmental conditions.
Figure 3 shows the variations in the relative abundances of alkylated imidazole homologs in the meteorite extracts with respect to the number of carbon atoms in the alkyl groups under the above assumptions. The variation in the Tagish Lake extract is clearly distinct from that in the other two meteorite extracts, suggesting a different parent body process. In contrast to the predominance of imidazole and its homologs, pyrazole (Supplementary Fig. 1), the structural isomer of imidazole, was not detected in any of the meteorite extracts. Laboratory experiments by Vinogradff et al.39 on the hydrothermal degradation of HMT in the presence of phyllosilicates (~150 °C for up to 31 days) provided various alkylated pyrazoles. We observed several unidentified peaks for the structural isomers of the alkylated imidazole and/or pyrazole (Supplementary Fig. 7), implying that alkylated pyrazole homologs were also present in the meteorite extracts. We also tentatively observed N-containing compounds with a linear structure (see Supplementary Text and Supplementary Fig. 8), where the hypothesized concentration trend between meteorites follows the same trend as that of alkylimidazoles (Supplementary Fig. 9), suggesting a distinct molecular distribution between the Tagish Lake meteorite and the other two meteorites.
Fig. 3: Possible relative abundances of alkylated imidazole analogs. Variations in the hypothesized relative abundances of imidazole and its alkylated homologs in the Murchison #1, Tagish Lake, and Murray extracts, with relevance to the number of alkylcarbons as [(-CH 2 -)n]. The relative abundances of these molecules in the organic residues formed by photochemical reactions of interstellar ice analogs are shown for comparison. Full size image
Supplementary Fig. 10 shows mass chromatograms at m/z 123.0553 and 124.0393, which corresponds to those of the protonated ions of nicotinamide and nicotinic acid, respectively, in the Murchison #1 extract. Based on their chromatographic retention time and accurate mass measurements of their parent masses, we identified two molecules in each meteorite extract. Both molecules have a pyridine structure with a carboxyl or amide group (Supplementary Fig. 10) and are generally classified as vitamin B 3 . Nicotinic acid has been identified in various carbonaceous meteorites with concentrations on the order of hundreds of ppb40, which is consistent with the results of the present study (Table 2). On the other hand, nicotinamide was not found in the hot water extract (at 100 °C for 24 h), acid-hydrolyzed hot water extract, or formic acid extract40. Under these severe extraction conditions, nicotinamide could have been hydrolyzed to yield nicotinic acid. Because the extraction method we used in our study was much gentler, as explained in the methods section, we believe that it may be more suitable for extracting such an acid-labile molecule. Supplementary Table 2 shows a relative abundance of nicotinic acid and nicotinamide (acid/amide) in each meteorite extract, as well as that value in the laboratory-made photochemical products prepared in our previous study25. Nicotinic acid was more abundant than nicotinamide in all meteorite extracts, but the degree varied between meteorites. For both Murchison and Murray extracts, the acid/amide ratio was ~10, whereas it was ~20 for the Tagish Lake extract. Since Tagish Lake meteorites are considered to have experienced a range of aqueous alteration conditions on the parent body41, the high acid/amide ratio may be indicative of the parent body processes. In fact, the acid/amide ratio for the photochemical products, which did not undergo any heating processes25, showed an acid/amide value of 0.8, supporting the above assumption. Further experiments, such as hydrolysis of nicotinamide, may be helpful to better understand the potential of the nicotinic acid/nicotinamide ratio for estimating the degree of aqueous alteration in meteorite parent bodies. In addition to nicotinic acid and nicotinamide, their structural isomers, such as isonicotinic acid and isonicotinamide, respectively, were identified in the same mass chromatograms (Supplementary Fig. 10), which indicates a wide diversity of organic molecules in the meteorite extracts, as described in the next section.
Wider diversity of pyrimidine molecules compared to previous studies
The detection of diverse suites of pyrimidine nucleobases and their analogs is distinct from previous reports on the detection of nucleobases in carbonaceous meteorites (ref. 11 and references therein). One possible reason for this might be the sample heterogeneity between the Murchison #1 and Murchison #2 meteorites (Table 2). However, the observed heterogeneity mainly arises from the concentration of the detected pyrimidine molecules rather than molecular diversity. We are surely certain that analytical and instrumental developments applied in the present study have a significant advantage for detecting ppb to ppt (= nanogram to picogram per gram-meteorite) orders of pyrimidine molecules in meteorite extracts. For example, ion-exchange chromatography as a desalting process improved after Takano et al.42 should have resulted in the higher detectability of these molecules by removing coexisting molecules that potentially interfere with the detection of pyrimidine molecules25. In addition, the HPLC conditions (e.g., the use of formic acid in the HPLC solvent as a protonated reagent), which are distinct from those applied in previous studies (for example, ref. 43) may have enhanced the detectability of these molecules. In fact, under the present analytical conditions, even for sub-nanogram-order nucleobases, their peaks are outstanding (Figs. 1 and 2), which indicates a much higher detectability of nucleobases.
Possible contribution from photochemical reactions in the ISM to the distribution of N-heterocyclic molecules in meteorites
The above discussion provides robust evidence that the nucleobases and their analogs detected in meteorite extracts are of exogenous origin with a unique molecular history. We previously demonstrated that pyrimidine and purine nucleobases can be formed by photochemical reactions of ice mixtures containing water, carbon monoxide, methanol, and ammonia under interstellar medium (ISM) conditions25. Although the detailed formation pathways by photochemical reactions are currently not well characterized, formamide (NH 2 CHO) is also a possible source of nucleobase formation in extraterrestrial environments22,25. It is noteworthy that photochemical reactions of interstellar ice analogs yield pyrimidine nucleobases one order of magnitude more abundant than purine nucleobases, but even pyrimidine nucleobases are minor compared to their structural isomers in the product25. These molecular properties of photochemical products (Supplementary Table 3) closely resemble those of the Tagish Lake extract, as shown by the high relative abundance of pyrimidine molecules compared to purine ones (Supplementary Fig. 11). In addition, the possible distributions of alkylated imidazole homologs in the Tagish Lake extract were similar to those of photochemical products prepared in previous studies (Fig. 3)25. These similarities imply that photochemical reactions in the ISM may partly contribute to the presence of nucleobases and related molecules in the Tagish Lake meteorite. An interstellar origin of meteoritic organic molecules was also inferred for HMT detected in the Murchison, Tagish Lake, and Murray meteorites26.
It is highly probable that, in addition to the pathways proposed above, there should be other routes to the formation of purine and pyrimidine nucleobases present in carbonaceous meteorites (see Supplementary Text). In addition, nucleobases with amino groups may easily decompose by hydrothermal activities on the meteorite parent bodies, and some molecules yield their oxygenated counterparts (e.g., from cytosine to uracil)33. A full understanding of the formation pathways of purine and pyrimidine nucleobases in carbonaceous meteorites is beyond the scope of the present study. Nevertheless, we believe that the detection of various molecules capable of acting as intermediates or precursors for nucleobase synthesis provides a significant advance in our understanding of prebiotic chemistry in meteorites.
Future perspectives and possible relevance to the origin of life on the early Earth
We reported the detection of more than 30 nitrogen heterocyclic molecules, including several DNA/RNA nucleobases in the meteorite extracts; however, a number of related molecules remain unidentified in the meteorite extracts (e.g. Supplementary Fig. 3c). Bulk analysis of high-resolution mass spectra of meteorite extracts by computational operations such as ATTRIBUTOR software10 can help identify families of homologous compounds present in meteorite extracts and thus can be used on our target molecules. Moreover, purely computational approaches, such as ab initio molecular dynamics simulations, to search for chemical reaction pathways in a complex system, have been developed in the last decade44,45. Looking ahead to the next decade in combination with laboratory-based analysis and model simulation studies, we are now approaching a more complete understanding of the molecular distributions of meteoritic organics, including nucleobases and their formation pathways. In this context, it is highly anticipated that various kinds of nucleobases and their analogs can be detected from the samples returned from the carbonaceous asteroids Ryugu1,46,47 and Bennu2.