Participants
For the current study, from a cohort of n = 1927, we selected an age and sex-matched sample of N = 440 with n = 110 acute, chronic, and remitted participants per group of MDD, BD, SSD, and HC (matched using the MatchIt package [26] in R [27]) (see Table 1 for descriptive statistics). As the SSD group was the smallest (n = 110), MDD and BD patients and HCs were matched 1:1 to the age and sex-distribution of the SSD group. Participants aged 18–65 were drawn from the Marburg-Münster Affective Disorder Cohort Study (MACS). MACS is a longitudinal bi-center cohort study, and part of FOR2107, a consortium investigating the neurobiology of major psychiatric disorders [28].
Table 1 Descriptive statistics. Full size table
Participants were recruited from in and out-patient departments of the universities of Marburg and Münster, Germany, local psychiatric hospitals (Vitos Marburg, Gießen, Herborn, and Haina, LWL Münster, Germany), and via postings in local newspapers and flyers. During a semi-structured interview, the German version of the structured clinical interview (SCID-I) for the DSM-IV-TR was applied by trained staff [29]. Furthermore, within one week of MRI scanning, psychopathological scales, a large questionnaire battery, a neuropsychological test battery and other rater-based scales were applied (see below). Moreover, clinical variables such as age of onset and number of hospitalizations were assessed during the semi-structured interview and, if available, based on patient records.
Exclusion criteria were a history of neurological or general medical conditions, current or lifetime alcohol dependency, current substance dependency, or current use of benzodiazepines (all assessed during the semi-structured interview performed by trained personnel and via self-report questionnaires), and verbal IQ ≤ 80 (assessed using [30]). Further exclusion criteria for the HC group were current or past mental disorders according to DSM-IV-TR, and lifetime intake of psychotropic medication. The study protocols were approved by the local Ethics Committees of Marburg and Münster, Germany according to the Declaration of Helsinki. All participants gave written informed consent before participation and received financial compensation.
Transdiagnostic phenotypical domains
To associate GMV with phenotype and risk factors, four domains were investigated: (1) early risk and protective factors, (2) current risk and protective factors, (3) psychopathology, (4) neuropsychological performance.
Early risk and protective factors
Childhood maltreatment was assessed using the childhood trauma questionnaire (CTQ) [31], urbanicity was assessed using a score for urban upbringing [32]. Familial risk was assessed via a questionnaire, asking whether a first-degree relative had been diagnosed and/or treated for MDD, BD, or SSD. Gestational age and birth weight were assessed through a questionnaire. Maternal and paternal care as an early protective factor was assessed with the German version of the parental bonding instrument (PBI) [33, 34].
Current risk and protective factors
Stressful life events (during the last six months) and their subjective impact were assessed using the life event questionnaire (LEQ) [35]. Current protective factors such as social support and resilience were assessed using the social support questionnaire (FSozu) [36] and the RS-25 resilience questionnaire, respectively [37].
Psychopathology
Psychopathology was assessed using the Scale for the Assessment of Negative Symptoms (SANS) [38], the Scale for the Assessment of Positive Symptoms (SAPS) [39], the Hamilton Depression Scale (HAM-D) [40], the Hamilton Anxiety Scale (HAMA) [41], and the Young Mania Rating Scale (YMRS) [42]. Further, the Global Assessment of Functioning (GAF) score [43, 44], was applied. All scales were rater-based. Raters were trained in the evaluation of psychopathological symptoms. Interrater reliabilities (ICC) were >0.86 for all scales.
Neuropsychological performance
A comprehensive neuropsychological test battery was applied including the d2 attention test [45], verbal fluency [46], symbol-coding [47], spatial span [48], letter-number span [49], Trail-Making Test A and B (TMT) [50], and the German Verbal Learning and Memory Test (VLMT) [51].
MRI data acquisition and preprocessing
MRI data acquisition and preprocessing was performed according to standardized procedures and using default parameters implemented in the respective toolboxes. At both sites, a 3-T MRI scanner (Marburg: Tim Trio, Siemens, Germany; Münster: Prisma, Siemens, Germany) was used to acquire T1 weighted images using a fast gradient echo MP-RAGE sequence with a slice thickness of 1.0 mm and a field of view of 256 mm. In Marburg, a 12-channel head matrix Rx-coil was used, in Münster, a 20-channel head matrix Rx-coil was used. Parameters differed across sites: Marburg: 176 sagittal slices, time of repetition (TR) = 1.9 s, time of echo (TE) = 2.26 ms, inversion time (TI) = 900 ms, flip angle = 9°; Münster: 192 sagittal slices, TR = 2.13 s, TE = 2.28 ms, TI = 900 ms, flip angle = 8°. MRI data were acquired according to an extensive quality assurance protocol [52].
A senior clinician visually inspected all scans regarding artifacts and anatomical abnormalities before preprocessing. Structural MRI data were preprocessed with the CAT12-Toolbox (Computational Anatomy Toolbox for SPM, build 1720, Structural Brain Mapping group, Jena University Hospital, Germany) (http://dbm.neuro.uni-jena.de/cat/) building on SPM12 (Statistical Parametric Mapping, Institute of Neurology, London, UK) using default parameters. In short, preprocessing included image segmentation into gray matter, white matter, and cerebrospinal fluid [53], spatial registration, and normalization [54]. Data were normalized to Montreal Neurological Institute (MNI) space. A more detailed description of our quality protocol can be found elsewhere [52]. During preprocessing, total intracranial volume (TIV) was calculated. MRI data sets were spatially smoothed with a Gaussian kernel of 8 mm full width half maximum.
Statistical analyses
Factor analysis of neuropsychological tests
Using SPSS 27, (Statistical Package for Social Science, IBM) an explorative principal axis factor analysis with varimax rotation was performed to identify aggregated domains of cognitive functioning (see Supplement). As neuropsychological test variables were differentially scaled, z-transformed values were used for the factor analysis. Suitability of neuropsychological test data for factor analysis was tested using Bartlett’s test of sphericity [55] and the Kaiser–Meyer–Olkin test (KMO) [56]. Factors were extracted according to the Kaiser’s eigenvalue greater than one criterion [57]. Factor loadings were extracted using the regression method as implemented in SPSS.
Brain structural analysis
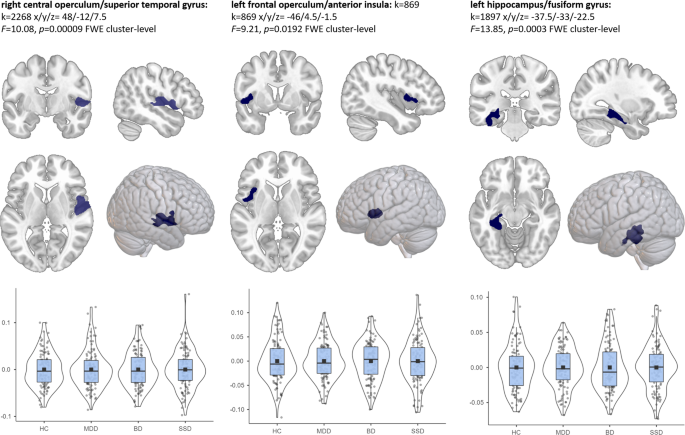
To identify differences across the four groups (HC, MDD, BD, SSD), smoothed GMVs were applied using a whole brain 1×4 design in SPM (v6906) running under Matlab (R2017a). Post-hoc tests between two respective groups were performed to investigate significant differences. To identify commonly altered areas across the three diagnoses vs. healthy participants, we performed a conjunction analysis. Conjunction analysis allows the identification of overlap in GMV alterations in single diagnosis vs. control comparisons. The conjunction analysis used here was defined as follows: HC > MDD ∩ HC > BD ∩ HC > SSD. As conjunction analysis is known to be conservative, we opted for a region of interest (ROI)-based approach which included previously identified ROIs reported in the meta-analysis [2] in MDD, BD, and SSD patients by Goodkind et al. (2015) [2]. These ROIs were the bilateral insulae, thalamus, ACC, left amygdala, and left hippocampus. Using the Dartel space neuromorphometrics atlas in CAT12, we created one mask that included these anatomical ROIs. We have also included an exploratory whole-brain conjunction analysis in the Supplement.
The covariates age, sex, and total intracranial volume (TIV) were used in all analyses. MRI data were acquired at two sites. As recommended by the MRI quality assurance protocol of the FOR2107 cohort, we used two dummy-coded variables accounting for the change of a body coil and site (Marburg pre body coil: yes/no, Marburg post body coil: yes/no, and Münster as reference category [52]). Following the CAT12 recommendations (http://dbm.neuro.uni-jena.de/cat/), threshold masking with a value of 0.1 was applied for all analyses to exclude non-brain areas. Results were considered significant at p < 0.05 cluster-level family wise error-corrected (FWE) for multiple comparisons after an initial threshold of p < 0.001 uncorrected, with a cluster extend threshold of k > 10. Significant clusters were labelled using the Dartel space Neuromorphometrics atlas (http://www.neuromorphometrics.com/).
Transdiagnostic phenotypical factors associated with GMV
To answer the question which factors were associated with transdiagnostic morphometric findings, we associated the identified conjunction cluster with four different domains. These were (1) early risk and protective factors, (2) current risk and protective factors, (3) psychopathology and (4) neuropsychology (see above). To this end, we extracted eigenvariate values (weighted means) as an approximation of mean value from the conjunction cluster. Using SPSS 27, we ran four separate multiple regression models. We decided against one combined model, as this would have resulted in a largely reduced sample size. Using four models allowed us to minimize sample size loss without having to impute values. In all models, age, sex, TIV, two site/bodycoil variables, and a group variable were used as covariates. Multicollinearity in these variables was absent.
The first model, early risk and protective factors, included childhood maltreatment, urbanicity, parental bonding, gestational age, birth weight, and familial risk. The second model, current risk and protective factors, included current life events, resilience, and social support. The third model, psychopathology, was comprised of global functioning, positive, negative, depressive, and manic symptomatology. The fourth model, neuropsychological performance, included three factors identified in the factor analysis described in the Supplement.