This is an initial report describing the presence of SARS-CoV-2 RNA and protein in the stool of 14 preterm newborns during hospital admission at delivery with negative SARS CoV-2 nasal PCR results and born to mothers with COVID-19 infection during pregnancy. The Spike protein, a SARS-CoV-2 surface protein that mediates viral entry, was consistently detected at high levels in one-third of infants.
Notably, maternal COVID-19 infection in our study was at least 10 weeks remote from delivery (Fig. 2c and Table 1), and 11 out of 13 mothers in this cohort had no active COVID-19 symptoms at delivery, making direct contact, droplet, or airborne transmission unlikely. Stool was collected as early as the first day of life. This suggests in utero SARS-CoV-2 transmission to the fetal intestine as a mechanism for the virus to be transmitted to the newborns. Preterm twins (P13 and P14), born to a mother who delivered in the adult intensive care unit due to severe COVID-19, showed no detectable viral RNA or protein in their stool (Fig. 2c and Table 1). We speculate that in utero transmission in this case may have been unlikely at 31 weeks GA due to more developed mechanisms to protect the fetus. Earlier pregnancy is associated with higher placental expression of viral entry receptors, including angiotensin-converting enzyme 212 and neuropilin.13 This suggests that in utero transmission of SARS-CoV-2, if possible, may be more likely during earlier gestion.
The mechanism of transmission to the fetal intestine remains unclear. In utero transmission of viruses, such as CMV, is transplacental. Additionally, SARS-CoV-2 has been detected in placentas.13,14 Viral RNAs were detected in the placentas of two mothers in this cohort with active COVID-19 at delivery (Table 1). SARS-CoV-2 viral RNAs have also been detected in amniotic fluid,15 which may be ingested by the fetus and passed through to the fetal intestine in utero.
Currently, our understanding of the impact of antenatal COVID-19 on the newborn is limited. SARS CoV-2 nasal PCRs performed in the immediate post-natal period of this cohort of newborns born to mothers with COVID-19 during gestation at our institution have been negative at the time of data analysis.16 However, negative nasal PCRs do not exclude the possibility of SARS-CoV-2 present in other tissues of the newborn, which have not been rigorously studied. A recent study by Boateng et al. reported no detection of SARS-CoV-2 viral RNA in the stool and urine from term newborns whose mothers had COVID-19 during pregnancy.17 However, the newborns in this study by Boateng et al. were term or near term (average GA ~39 weeks), compared to the average GA of 34 weeks in our NICU cohort. Our findings suggest increased risk of in utero transmission when maternal COVID-19 occurred prior to ~27 weeks of gestation. Furthermore, all stool and urine samples were collected at 15 h following birth in the study by Boateng et al.; viral titers might be below the detection limit at such an early time. In contrast, many of our samples were tested at multiple time points up to two months of age. Therefore, there are inherent differences in the infant cohorts and timing of fecal sample collection between our study and the study by Boateng et al.
Another study by Zeng et al.5 describes 33 neonates, including 30 with negative nasopharyngeal and anal PCR swabs born to mothers with infection at various time points during pregnancy. 3 neonates were diagnosed with active COVID-19 with positive nasopharyngeal and anal swabs, and all 3 were born to mothers diagnosed with active infection at delivery. The negative anal swabs described may only have reflected the lack of RNA present on the skin’s surface and may have been taken prior to any stooling. In contrast, our study directly assessed the presence of both SARS-CoV-2 viral RNA and protein in newborn stool reflective of intestinal lumen contents. The majority of our infants (11 of 14) were born to mothers with COVID-19 resolved more than 10 weeks prior to delivery; all of these 11 infants had SARS-CoV-2 RNA and Spike protein detected in the stool.
Persistent viral reservoirs in the intestine have been found in both recovered adult and pediatric COVID-19 patients 4 months after infection, suggesting the intestinal environment providing a unique niche for prolonged viral reservoirs.18,19 Enterocytes and smooth muscle cells in the intestine express high levels of ACE2 and neuropilin, respectively.20,21 A recent study demonstrates high expression of ACE2 in the human fetal intestine in early second trimester.22 Additionally, the intestinal lumen is less accessible to immune cells. These factors may collectively create a niche in the intestinal lumen that is conducive to the establishment of what may be persistent viral reservoirs of SARS-CoV-2 in the fetus and newborn.
The clinical implications of our findings of viral RNAs and Spike protein in the stool of these newborns require further investigation. Most newborns studied here were either clinically well or progressed as expected during hospital admission for their gestational ages. There were two symptomatic cases presented in this report. While there is insufficient evidence to connect the development of symptoms to effects of COVID-19, It remains unclear if the presence of intestinal reservoirs of SARS-CoV-2 might have contributed to the development of GALD or NEC in these two newborns. Maternal viral infections known to cause maternal immune activation, including herpes simplex virus, enterovirus and cytomegalovirus, have been associated with the development of acute liver failure in newborns, which is commonly caused by GALD.23 The immunopathology of COVID-19 involves excessive inflammatory response, impaired adaptive immunity, and production of autoantibodies.24,25,26 Infections during pregnancy may increase the risk of maternal alloimmune responses, such as GALD, whereby maternal IgG antibodies attack fetal hepatocyte antigens, resulting in fetal and neonatal liver injury. Autoantibodies that antagonize type I IFN responses were found in 19% of patients with critical COVID-19.27 Autoantibody reactivities in pregnant women with COVID-19 may present potential additional risk to the fetus, as maternal autoantibodies unleashed by SARS-CoV-2 infection may attack fetal cells. Further investigation is required to elucidate autoantibody responses in pregnant women with COVID-19 and possible risk to the fetus.
The case of NEC raises the concern that intestinal inflammation driven by an intestinal reservoir of SARS-CoV-2 may increase the risk of gastrointestinal complications in these newborns, such as NEC. COVID-19 has previously been implicated in the development of NEC in newborns.28,29 However, NEC may have developed irrespective of maternal COVID-19 status in this newborn who was already at high risk due to prematurity.
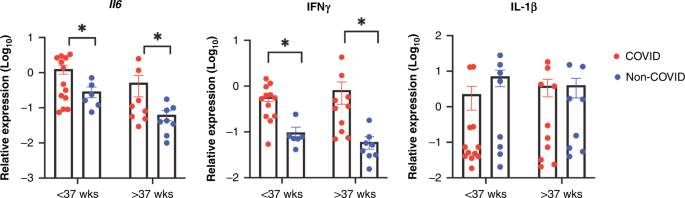
IL-6 and IFN-γ are both proinflammatory cytokines that have been reported to be elevated in severe COVID-19 patients.30,31 In particular, as a potent innate cytokine, IL-6 is thought to contribute to potentially fatal cytokine storms in COVID-19.30,32 Increased induction of these cytokines in the neonatal intestine by viral RNAs may impact the immune cell development and immune landscape in the neonatal intestine and may potentially affect disease susceptibility in later life.
Limitations of this study include a small sample size. Control group newborns born to mothers without a confirmed or reported case of COVID-19 during pregnancy were limited to newborns admitted at the time of our study. Furthermore, the current understanding is that persistent intestinal reservoirs of SARS-CoV-2 may not reflect an active infection.33
It is unclear if the presence of viral RNA and protein within the gut microbiome represents active virus in newborns with clinical hospital courses typical of their gestational age in 12 out of 14 cases. However, increasing levels of viral RNA and protein over time suggest replication in some infants. The stool homogenates from the COVID-19 cohort induced a significant increase in IL-6 and IFN-γ production in macrophages, reflecting higher amounts of inflammatory components in the intestinal lumen of these infants, consistent with the detection of SARS-CoV-2 viral RNA and protein in their stool specimens. Early development of the immune system is heavily influenced by the gut microbiome;34 the full impact of persistent viral reservoirs on the development of the immune system in these infants may not be fully appreciated until years later. Further investigation is required to understand the mechanisms of in utero transmission of SARS-CoV-2 in women with COVID-19 during early stages of gestation, as well as the full impact of persistent intestinal viral reservoirs on child health and development.