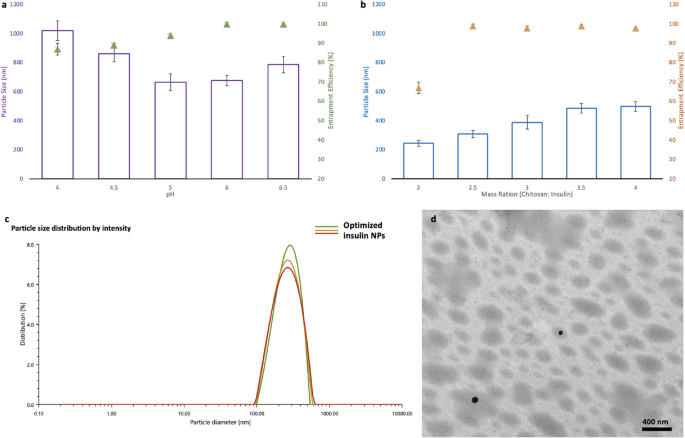
Optimization of different parameters to generate homogenized insulin NPs
The pH of the mixing solution and the ratio of the chitosan and insulin were two critical factors affecting the particle size and encapsulation efficiency (EE) of the final NPs, as they directly influenced the ionotropic gelation process15. The pH of the mixing solution was shown to have high associations with both particle size and encapsulation efficiency (Fig. 1a). As shown in Fig. 1a, the mean particle size (nm) dropped, and EE increased remarkably with pH increased from 4.0 to 6.0, while the mean particle size started to increase and the EE remained the same once the pH increased to 6.5. With increasing the ratio of chitosan to insulin, the mean particle size kept increasing as well. Besides, no change in the EE was observed when the nanoparticles were prepared under a chitosan/insulin mass ratio higher than 2.5:1 (w/w) (Fig. 1b). Accordingly, the optimal preparing condition in this research (pH 6.0 and chitosan/insulin mass ratio of 2.5:1) was used to prepare insulin-loaded nanoparticles for further studies. With this preparation condition, the insulin NPs were optimized to 318 nm of mean particle size (Fig. 1c), 0.18 of PDI, 99.4% of entrapment efficiency, 9.8 mv of zeta potential 25.01% (m/m) of insulin loading content. Based on the transmission electron microscope (TEM) results, the optimized nanoparticles were roughly spherical and discrete with relatively uniform size (Fig. 1d).
Figure 1 Optimization of parameters for insulin NPs: (a) Effect of the pH on the average diameter and the encapsulation efficiency (EE) of the insulin NPs (prepared under the mass ratio of chitosan and insulin at 5:1); (b) Effect of the mass ratio between chitosan and insulin the average diameter and the encapsulation efficiency (EE) of the insulin NPs (prepared at pH 6); (c) Particle size distribution of the optimized insulin NPs; (d) TEM micrographs of the optimized insulin NPs. Full size image
It is well known that chitosan is a weak polyelectrolyte with a pKa of 6.5. It is positively charged in acidic media because its main amino groups are protonated by hydrogen ions15. It is, therefore, frequently utilized as a carrier for encapsulation of negatively charged macromolecules. In this study, chitosan was used to encapsulate insulin with an isoelectric point of 5.3. Since the chitosan served as a coating material, with the increase of its ratio, the thickness of the outer layer of the NPs increased accordingly, resulting in a larger mean particle size. Additionally, higher content of chitosan can encapsulate more insulin. In our case, the highest EE was achieved once the ratio of chitosan and insulin reached 2.5:1, and no significant change in the EE was observed when the ratio kept increasing.
Apart from the chitosan and insulin ratio, pH also played an essential role in NPs preparation. Gan et al.17 investigated the influence of pH on chitosan NPs particle size. They found that the particle size showed a continuous decrease before the pH reached the value of 6.0, and a significant increase in size was observed at pH > 6.0, which agrees with our observations. This phenomenon is because as pH rises, insulin molecules gain a negative surface charge, thus, favoring electrostatic interactions with the chitosan/ Sodium tripolyphosphate (TPP) complex and resulting in small particle sizes and high EE. However, when pH was tuned to 6.5, deprotonation of amino groups on chitosan occurred, leading to the folding of chitosan18. Thus, high pH caused fewer amino ions to be exposed to TPP and insulin, resulting in low crosslinking and eventually larger mean particle size and low EE.
Characterization of the dehydration NPs
Morphology analysis
Analyzing the morphological properties of freeze-dried and spray-dried NPs can guide selecting better dehydration and powder formation techniques. The preferred approach should provide drug stability, uniform particles shape, high drug loading, and good solubility in the original solution. In this study, to better compare the two techniques, insulin NPs with or without 1% of mannitol were used for the dehydration process. Mannitol was used as a bulking agent or cryoprotectants in various dry powder formulations for freeze-drying and spray drying. For the freeze-dried insulin NPs with no mannitol, as shown in Fig. 2a, a highly porous powder structure with large, irregular, and rough surfaces was observed under the scanning electron microscope (SEM). Almost no discrete particles could be detected in the powder after dehydration (Fig. 2e). These results indicated that most NPs were broken down during freeze-drying without any cryoprotectants. For both freeze-dried and spray-dried insulin NPs with 1% of mannitol, a spherical shape with a smooth surface of NPs was observed (Fig. 2b, d, f, h). The insulin NPs spray-dried with no mannitol kept their spherical shape but had a wrinkled surface (Fig. 2c). The spherical and wrinkled surfaces were further discussed in the following release behavior and cell uptake tests. Based on the visible appearance of the dry NPs, the NPs spray-dried with no mannitol and NPs freeze-dried and spray-dried with mannitol were all resulted in fine NPs powders (Fig. 2f, g, h). Higher surface area between the surface of microparticles induces a higher solubility and, thus, a higher release rate19.
Figure 2 Morphology of different dehydrated insulin NPs: (a) SEM image of freeze-dried insulin NPs without mannitol; (b) SEM image of freeze-dried insulin NPs with mannitol; (c) SEM image of spray-dried insulin NPs without mannitol; (d) SEM image of spray-dried insulin NPs with mannitol; (e) Image of freeze-dried insulin NPs powder without mannitol; (f) image of freeze-dried insulin NPs with mannitol; (g) Image of spray-dried insulin NPs powder without mannitol; (h) Image of spray-dried insulin NPs powder with mannitol. Full size image
During freeze-drying, mannitol acted as a cryoprotection agent, remaining in amorphous forms and preventing the NPs from damage by the ice crystal19. In contrast, there is no freezing step during the spray drying process. The mannitol was therefore not necessary in this method. In fact, as discussed earlier, spray-dried NPs without mannitol produced finer NPs. However, the mannitol could still serve as a bulking agent in the spray drying process to impart NPs a more spherical structure20 (Fig. 2d), which could help obtain a uniform release behavior of such encapsulated NPs. Moreover, it was evident that some large particles can be detected in both freeze-dried and spray-dried insulin NPs with mannitol (Fig. 2b, d), which probably resulted from the accumulation of mannitol in the particle's core alongside insulin inside the encapsulating layer of chitosan. Noticeably, in this study, to make sure that the spherical structure can remain intact after the dehydration, the ratio of mannitol and chitosan was kept at 5:1 so that the high amount of bulking agent can also enlarge the particle size of the dry NPs.
FTIR analysis
Fourier Transform Infrared-attenuated total reflectance (FTIR-ATR) spectroscopy characterized free insulin, chitosan, a physical mixture of chitosan, TPP, and insulin. All dehydrated NPs were characterized by using FTIR-ATR spectroscopy. Noticeably, band intensities at 1641, 1543, and 1412 cm−1 were observed in encapsulated NPs freeze-dried with mannitol and NPs spray-dried both with and without mannitol (Fig. 3). These increases in intensities are associated with the cross-link among chitosan, TPP, and insulin, as previously reported21. Studying the interaction between chitosan and insulin indicated that, in the FTIR spectra of insulin-loaded chitosan NPs, the chitosan bands overlapped with those of insulin, increasing the carbonyl intensity (1641 cm−1) and amine (1543 cm−1) bands. The tripolyphosphate groups of TPP linked with ammonium groups in chitosan caused the band at 1412 cm−1.
Figure 3 FTIR-ATR spectra of free insulin, chitosan, physical mixture of chitosan/TPP/insulin and NPs dehydrated by different methods. Full size image
Furthermore, these results were consistent with the results shown in SEM, which indicated the encapsulated NPs remained intact for both spray and freeze-drying with mannitol, but without mannitol, only spray drying could yield encapsulated particles. In contrast, the FTIR-ATR spectra result of the NPs freeze-dried without mannitol was very similar to the physical mixture of chitosan, TPP and insulin. This result indicated that cross-link among chitosan, TPP, and insulin no longer existed in NPs freeze-dried without mannitol. The NPs structure was broken during freeze-drying without cryoprotectant, which could be seen in the SEM result (Fig. 2a). Based on the morphological and FTIR results of the dehydrated insulin NPs, as the NPs freeze-dried with no mannitol broke down during the dehydration process, only NPs freeze-dried with mannitol, spray-dried with and without mannitol were used for reconstitution tests and discussion.
Reconstitution and stability analysis
Dehydration is being used for long-term storage and reprocessing to other formulations. The reconstitution ability of the dry NPs after the storage is critical for their use in different formulations such as tablets and films. We noticed that the mean particle size of the insulin NPs spray-dried with no mannitol was only slightly increased after reconstitution. On the other hand, the size of particles of insulin NPs spray-dried and freeze-dried with mannitol increased significantly (Table 1). The PDI and EE did not change substantially (p > 0.05) after the reconstitution for all NPs in this study (Table 1). This result indicated that most of the particles remained intact after redissolved. However, adding mannitol caused the insulin loading content of freeze-dried and spray-dried NPs with mannitol to decrease tremendously (Table 1). In contrast, the insulin loading content of NPs spray-dried without mannitol remained the same as before (Table 1).
Table 1 Physicochemical properties of freshly prepared and reconstituted NPs (one way ANOVA comparisons, p < 0.05). Full size table
It is well known that nanoparticles’ loading content is crucial when applied for drug delivery purposes. For NPs with low loading content, a very high quantity of material is required to achieve the therapeutic threshold. However, the high viscosity of such high NP concentrations leads to inconvenience and difficulties for oral delivery and injection formulations, respectively22. In addition, insulin NPs can also be used to make tablets and adhesive biofilms23,24, necessitating using a huge amount of low loading content NPs, resulting in large tablets and thick biofilms inappropriate for oral application. Accordingly, dehydrated NPs with high insulin loading are highly demanded. Our results suggested that the high insulin loading of the spray-dried NPs without mannitol could offer many attractive advantages to these alternative administration methods.
All dehydrated NPs were stored in the fridge for three months. The SEM result indicated that the morphology of all the dehydrated NPs had no visible changes during the three-month storage (Fig. 4). After reconstitution in water, The EE of all the NPs slightly decreased, and around a small amount (~5%) of insulin was released during the three-month storage (Table 2). However, the mean particle size of all the NPs increased. The particle size of the NPs spray dried without mannitol was increased to 525 nm while the particles size of the spray-dried and freeze-dried NPs with mannitol increased to 872 and 921 nm, respectively (Table 2).
Figure 4 Morphology of different dehydrated insulin NPs stored for three months: (a) SEM image of freeze-dried insulin NPs with mannitol; (b) SEM image of spray-dried insulin NPs without mannitol; (c) SEM image of spray-dried insulin NPs without mannitol. Full size image
Table 2 Physicochemical properties of NPs after three-month storage (one way ANOVA comparisons, p < 0.05). Full size table
Moreover, precipitates could be seen in the redissolved insulin NPs spray dried and freeze-dried with mannitol (Fig. S2). This was probably caused by the big particles, which could not suspend in water properly. All the results above showed that the spray drying technique can protect the insulin NPs from dehydration, and the high loading content of insulin NPs without any bulking agent or cryoprotectants could be obtained.
Insulin protection effects
Insulin retention ratio was tested in pH = 2.5 medium with pepsin, trypsin, and α -chymotrypsin to demonstrate the protection capacity of the NPs against enzymatic digestion after dehydration. The insulin retention ratio of the dehydrated NPs was compared to the freshly prepared NPs, and the free insulin was used as the negative control. In this study, free insulin showed fast insulin elimination within 4 h in all three enzyme treatments (Fig. 5a–c). In contrast, the insulin elimination test of the NPs freeze-dried with mannitol and the NPs spray-dried both with or without mannitol exhibited significantly higher protection effects of these NPs against enzyme digestion, which was similar to the freshly prepared insulin NPs (Fig. 5a–c). More than 50%, 60%, and 75% of the insulin can be protected within 4 h with the help of nanoparticles in pepsin, trypsin, and α -chymotrypsin, respectively (Fig. 5a–c). This insulin protection ability can increase the opportunity for higher insulin absorption into the blood circulation25. These results suggest that spray drying with or without mannitol and freeze-drying with mannitol can maintain the insulin protection ability of the NPs after dehydration.
Figure 5 Protection effects and release behaviors of dehydrated insulin NPs: (a) Protection of insulin at pepsin solution; (b) Protection of insulin at trypsin solution; (c) Protection of insulin at α-chymotrypsin solution; (d) Release behaviors of dehydrated NPs in pH = 2.5 solutions; (e) Release behaviors of dehydrated NPs in pH = 6.6 solutions; (f) Release behaviors of dehydrated NPs in pH = 7.0 solution. Full size image
In vitro release of insulin from NPs at different pH conditions
Freshly prepared and redissolved dry insulin NPs were incubated in various buffers (pH = 2.5, 6.6, 7.0) at 37 °C, modeling the pH environment in the stomach, duodenum, and upper small intestine, to examine the release behaviors of insulin in different segments of the GI tract. At pH = 2.5, insulin-loaded NPs and redissolved dry insulin NPs showed an initial burst release in the first one hour, followed by a slow release in the next 5 h (Fig. 5d). This fast release at the beginning was most probably the result of rapid surface desorption of protein molecules that were not fully entrenched in the particle's interior structure. At pH = 6.5, insulin-loaded NPs and redissolved dry insulin NPs showed a smoothly slow release in 6 h because the pH of the testing solution was similar to the NPs preparation solution (Fig. 5e). At pH = 7, NPs were unstable and almost fully disintegrated in the first two hours (Fig. 5f). This resulted from the fact that the deprotonation of chitosan happened at higher pH, which caused a less compact polymer network and the release of the loaded insulin.
Furthermore, the insulin NPs spray-dried with no mannitol showed a faster release profile than other dehydrated NPs (Fig. 5d–f). As previously discussed, the redissolved insulin NPs pray dried with no mannitol showed the smallest particle sizes. The small particles provided a large surface area so that most of the drugs associated would be at or close to the particle surface, resulting in fast drug release26.
In vitro toxicity evaluation
An MTT assay was employed to investigate the cytotoxicity of NPs. As shown in Fig. S4, all dehydrated NPs were found to have no significant impact on the cell viability at the concentration of 50–500 μg/ml, which indicated that all dehydrated NPs can be safely used to reach the therapeutic window.
In vitro cellular uptake of dehydrated NPs
The liver is the primary organ where insulin performs its physiological function. HepG2 cell is a human liver cancer cell line commonly utilized as a hepatocyte absorption model in vitro. Herein, HepG2 cell was used to evaluate the cellular uptake of NPs dehydrated using freeze-drying and spray-drying methods. After hours of incubation with free FITC-insulin concentration of 25 μg/mL, equivalent insulin concentrations of freshly prepared FITC-insulin loaded NPs, and dehydrated FITC-insulin loaded NPs, the cellular uptakes were quantified using flow cytometry and visually by confocal laser scanning microscopy (CLSM) observation. The freeze-dried NPs without mannitol were broken during the dehydration process and were not evaluated in this test. The intracellular fluorescence intensities (Fig. 6a) of freshly prepared insulin-loaded NPs, freeze-dried NPs with mannitol, and spray-dried NPs with and without mannitol were 4.3, 2.6, 2.4 and 4.1-fold higher than the intensity of the free FITC-insulin group, respectively (Fig. 6b). These results demonstrate the higher efficacy of encapsulated insulin than free insulin in cellular uptake, which is primarily associated with the smaller size of the insulin-loaded NPs created in the study.
Figure 6 HepG2 cellular uptakes after 4 h incubation with fresh prepared NPs and dehydrated NPs: (a) Distribution of FITC-insulin uptaken by HepG2 cells. (b) Geometric mean values of the fluorescence intensities of the flow cytometry analysis (n = 3), *P < 0.05 compared with free insulin. Full size image
Similarly, the CLSM images showed that the FITC fluorescence intensity of fresh prepared FITC-insulin loaded NPs and FITC-insulin loaded spray-dried NPs without mannitol was much stronger than the intensities of the other samples (Fig. 6a). Besides, with the addition of mannitol, the higher viscosity of the solution increased the resistance of cellular uptake27, resulting in lower insulin proliferation. These results indicate that spray-dried NPs without mannitol demonstrated the highest cellular uptake efficiency, as their particle sizes were smaller than freeze-dried NPs once redissolved.