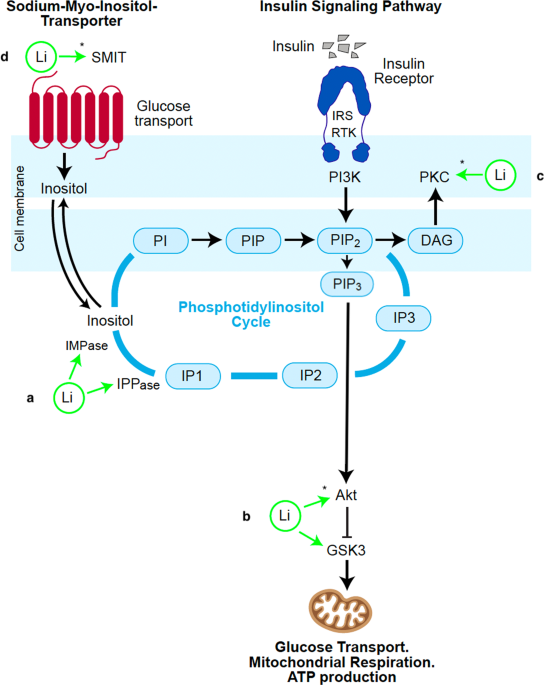
Sodium myo-inositol transporters (SMIT) are a proposed, indirect target of lithium. SMIT determines the level of available inositol in the cell to act as a substrate for insulin signaling and therefore plays an important role in metabolic function. A study on SMIT mRNA in BD reported a significantly higher expression of SMIT mRNA in untreated bipolar type 1 patients and that this was downregulated by treatment with lithium [60]. Upregulation of SMIT may reflect an increased demand for inositol due to hyperactive PI-cycle activity in bipolar type 1 patients who experience mania more frequently than those with type 2 BD. In the same study, expression of SMIT mRNA was not found to be increased in bipolar type 2 patients who experience depression more frequently. This may provide an indication that the manic state increases the demand for inositol to facilitate a hypermetabolic state. Lithium acting to inhibit hyperactivity of the PI-cycle would then necessarily reduce the demand for SMIT transport of inositol. Lithium uncompetitively inhibits IMPase and IPPase in the phosphatidylinositol cycle and so does not inhibit inositol metabolism unless it becomes active above a certain threshold [15]. Therefore, lithium may have little effect on states of depleted intracellular inositol but prove to be very effective in states of overactivity of the inositol cycle. This explanation is consistent with the stronger anti-manic than antidepressant effect of lithium. The uncompetitive inhibition lithium exerts has been noted in the literature to be an advantage over PI-cycle inhibitors which act indiscriminately to supress PI-cycle activity [51].
Glucose and inositol are transported into the cell by SMIT and hyperglycemia has been shown to inhibit SMIT uptake of inositol [61]. Additionally, hyperglycemia inhibits re-uptake of myo-inositol in the kidney and increases urinary excretion of myo-inositol. Excretion of myo-inositol is common in IR conditions and urinary detection of myo-inositol is used as a diagnostic tool [62]. Metabolomic evidence of altered inositol phosphate metabolism in urine samples from BD patients has been observed, although more data is needed to establish this [63]. In addition to preventing inositol from reaching the CNS, hyperglycemia and insulin resistance alter the ratio of inositol isomers in tissues. Detection of changes in these isomer ratios in blood and urine are used as early markers of insulin resistance in several metabolic conditions [64]. In conditions of insulin resistance such as polycystic ovary syndrome (with which BD is significantly comorbid) inositol supplementation is a standard treatment and has been demonstrated in several randomized controlled clinical trials to improve insulin sensitivity and lower blood glucose [65]. The leading hypothesis for this effect of inositol is that it acts to restore inositol substrate for PI3K/Akt insulin signaling [61]. A small trial of inositol for bipolar depression reported 50% or greater decreases in MADRS scores in 8 of the 12 patients taking inositol [66]. And reduced brain myo-inositol is also associated with major depressive disorder (MDD) [67], however, trials of inositol supplementation for MDD are inconclusive [68].
Acute experimental diabetes reduces the concentration of free myo-inositol in peripheral nerves in both animals and humans [69, 70]. Hyperglycemia and hyperinsulinemia-mediated inhibition of inositol uptake to the CNS may leave the CNS deprived of inositol thereby inhibiting insulin signaling. Barkai et al. report that CNS inositol levels were depleted in samples from hospitalized BD patients [71]. It may be expected that under such conditions the brain upregulates de-novo synthesis of inositol to compensate. A spectrophotometric study showed that G6PD which facilitates the de-novo synthesis of myo-inositol was positively correlated with a marker of mitochondrial dysfunction in BD. This result may indicate that G6PD-mediated inositol synthesis plays a role in a compensatory mechanism for mitochondrial dysfunction in BD [72]. There is limited research on SMIT and its interaction with lithium in BD and so only modest conclusions can be drawn at this time. However, the significant role of SMIT in facilitating PI-cycle activity and its relevance to insulin resistance provide further support for a metabolic component to lithium action.