Carbon monoxide, aerosol mass and water
Table 1 summarizes the per-puff levels of CO, aerosol collected mass (ACM), water and nicotine in the emissions from two e-cigarettes: namely, Berry Blast 57 mg mL−1 of nicotine containing lactic acid (BB57); and Berry Blast 18 mg mL−1 of nicotine containing benzoic acid (BB18). CO, which is associated with combustion of organic material, was below the limit of detection (< LOD) for both e-cigarettes, with a percentage reduction of 99.8% relative to Ky1R6F cigarette smoke (Table 1). ACM, which comprises mainly PG, VG, water, nicotine and other minor constituents, was in the same range for both e-cigarettes. ACM results were found to be reproducible across all methods as demonstrated by the low standard deviation of ACM in both e-cigarette emissions (6.58 ± 0.39 mg puff−1 and 6.46 ± 0.36 mg puff−1 for BB57 and BB18 respectively), accounting for a coefficient of variation of 5.9% and 5.5% for BB57 and BB18, respectively (n = 85). This is an indication of sampling robustness and puffing consistency. The nicotine-free dry particulate matter (NFDPM) or ‘tar’, a parameter associated with cigarette smoke, consists predominantly of combustion by-products36,58. The level of NFDPM, 3.67 ± 0.30 mg puff−1 equivalent to 33 ± 3 mg cig−1, was in accordance with the Ky1R6F certified value of 29 ± 2 mg cig−1 (ISO Intense smoking regime)56.
Table 1 CO, ACM, NFDPM, water, nicotine, humectants and related impurities and toxicants: per-puff concentration of e-cigarette vapour emissions, respective method air blanks and Ky1R6F cigarette smoke. Full size table
Humectants and related impurities
In terms of humectants, levels of PG and VG were higher in the e-cigarette emissions than in the Ky1R6F cigarette smoke (Table 1). Because PG and VG are the main constituents of e-liquids, these results were expected. Diethylene glycol (DEG) and ethylene glycol (EG), which may be present in e-liquids as impurities59,60, were < LOD in the e-cigarette emissions. These compounds were raised as a potential concern by the U.S FDA after reports of their detection in e-liquids53,54,61. EG is widely used as an anti-freeze agent and is associated with pronounced toxicological risks50. The US Pharmacopeia (USP) has set a limit for DEG and EG of 0.1% (1 mg g–1) in both PG and VG59,60,62. Their low levels in the e-cigarette emissions shows the importance of using pharmaceutical-grade PG and VG. Glycidol, which is listed as a probable carcinogen by the International Agency for Research on Cancer (IARC)63, was < LOD for both e-cigarette emissions and cigarette smoke.
Nicotine, nicotine-related impurities and TSNAs
As shown in Table 1, different nicotine concentrations were observed for the e-cigarette emissions and Ky1R6F cigarette smoke, with nicotine levels in cigarette smoke (0.23 mg puff–1) lying between those in the two e-cigarette emissions (BB18, 0.10 mg puff–1; BB57, 0.32 mg puff–1). The concentration of nicotine in the BB57 emissions relative to BB18 was three times greater and followed the three-fold increase in nicotine strength of the e-liquid. In the cigarette smoke, the measured nicotine concentration of 0.23 ± 0.01 mg puff–1 (Table 1), equivalent to 2.07 ± 0.09 mg cig−1, is in accordance with the Ky1R6F certificate value of 1.9 ± 0.1 mg cig−1 (ISO Intense smoking regime)56. Nicotine yields for different e-cigarette brands have been reported from 2 to 313 µg puff−1 while for conventional cigarettes smoke the values ranged from 170 to 232 µg puff−16,31,36,48,64.
Nicotine-related impurities were present mainly in cigarette smoke at a significantly higher level than in e-cigarette emissions (note that the percentage reduction of anabasine and nicotine-N-oxide was not calculated because these impurities were < LOD in cigarette smoke and the e-cigarette emissions). In general, the nicotine used in e-liquids is extracted from tobacco and may contain other minor related alkaloids as impurities64,65. Therefore, nicotine-related impurities might be expected in e-cigarettes emissions and are considered acceptable by the USP and European Pharmacopeia in standard nicotine used in e-liquids6,66,67,68. The USP requires single impurities to be less than 0.5% (5 mg g–1) of nicotine, and total impurities to be less than 1% (10 mg g–1)66. The European Pharmacopeia requires each of seven specified impurities (anabasine, anatabine, cotinine, myosmine, nicotine-N-oxide, β-nicotyrine, nornicotine; Table 1) to be below 0.3%, unspecified impurities to be no more than 0.1% each, and total impurities to be less than 0.8% of nicotine content6,66,67,68. In our study, all analysed nicotine-related impurities in the e-cigarette emissions were below the levels stated by the USP and European Pharmacopeia for e-liquids (Table 1). This is consistent with the fact that only nicotine of pharmaceutical-grade is used in the production of these e-liquids. β-Nicotyrine, a pyrolysis product of nicotine69, was present at the highest level in cigarette smoke (0.42 µg puff–1). The observed reduction of 98% per puff in the emissions of both e-liquids is a good indication that the heat generated in the device atomiser is not sufficient to thermally breakdown nicotine to β-nicotyrine.
Another class of nicotine-related HPHCs of concern are tobacco-specific nitrosamines (TSNAs): namely, NNN, NNK, nitrosoanabasine and nitrosoanatabine. These non-volatile compounds may be present in e-liquids as impurities from tobacco nicotine extraction and are important compounds associated with negative health effects of cigarette smoke34,70,71,72,73,74. Two of the reported TSNAs, namely NNN and NNK, are classified as carcinogens and included in U.S FDA’s HPHC lists that apply to cigarette smoke and electronic nicotine delivery systems (ENDS)39,53,75. NNN and NNK are also included in the nine WHO TobReg priority smoke toxicants55. In our study, the levels of all four TSNAs were < LOD for both e-cigarette emissions with a percentage reduction of ≥ 99.9% as compared with cigarette smoke (Table 1).
Polycyclic aromatic compounds
Another class of chemicals in cigarette smoke that poses health concerns are polycyclic aromatic hydrocarbons (PAHs), which are compounds with two or more fused benzenoid rings that are known for their carcinogenic and mutagenic properties76. The levels of PAHs in e-cigarette emissions were either < LOD or < LOQ (chrysene), except for naphthalene and pyrene (Table 2). Notably, these two compounds were higher than the limit of quantification (LOQ) in the method air blanks. Pyrene was in the same concentration in e-cigarettes as in the method air blank (0.1 ng puff–1). PAHs are present in the atmosphere as components of various dusts, tars, oils and engine exhaust gases72. The presence of pyrene in the e-cigarette aerosol is therefore most probably an artefact due to environmental contamination, as indicated by the method air blank.
Table 2 Polycyclic aromatic hydrocarbons: Per-puff concentration of e-cigarette vapour emissions, respective method air blanks and Ky1R6F cigarette smoke. Full size table
Levels of naphthalene were slightly higher than those of pyrene in e-cigarette emissions, while the respective air blank was lower. Nevertheless, it seems likely that these compounds were detected in e-cigarette emissions due to their presence as low-level contaminants in the background air, rather than originating from the vaping product. In terms of the levels detected, even if we assume a worst-case exposure of 300 puffs per day based on the million puff study (which reported a median use of 130 puffs day–1 and where 85% of users did not exceed 300 puffs day–177), a consumer’s daily exposure to each of these compounds would be less than 0.15 µg day–1, the toxicological threshold of concern for mutagenic compounds78,79. Furthermore, most PAHs, including naphthalene and pyrene, showed a percentage reduction in e-cigarette emissions of ≥ 99% versus Ky1R6F cigarette smoke, while indenol[1,2,3-cd]pyrene showed a reduction of 92.5% because it was also present at only low levels in cigarette smoke (0.02 ng puff–1).
Collectively, our findings are consistent with the knowledge that PAHs are primarily products of combustion. For PAHs present at very high concentrations in cigarette smoke, such as benzo[a]pyrene, chrysene and pyrene, the percentage reduction in e-cigarette emissions was > 99%. In particular, benzo[a]pyrene, which is included in the nine WHO TobReg priority smoke toxicants, was reduced by 99.7% in e-cigarette emissions as compared with the smoke from the reference cigarette.
Phenolic compounds and carbonyls
In cigarette smoke, the phenols of concern are catechol, m-cresol, p-cresol, o-cresol, hydroquinone, phenol and resorcinol (Table 3). They can be formed by the thermal degradation of tobacco leaf constituents such as lignin and chlorogenic acid71,72,80,81. Temperature is an important factor in the formation of phenolic compounds. Studies have reported that catechol and hydroquinone are formed in cigarette smoke at low temperatures (< 350 °C), while cresol, phenol and resorcinol are formed at temperatures of 350–600 °C81. In e-liquids, phenols and their precursors may be present as impurities derived from nicotine and may be transferred to the aerosol and inhaled by the vaper71,72. Phenols may also be formed upon vaporisation. Phenol emissions have been found to be independent of the nicotine benzoate concentration but significantly correlated with the PG/VG ratio. Emissions increased with power and puff duration, consistent with conditions that lead to a higher temperature and greater thermal degradation82. In our study, the levels of all seven phenols were < LOD in both e-cigarette emissions with a percentage reduction of ≥ 99% versus cigarette smoke (Table 3). The low operating temperatures of the e-cigarette device studied herein and the use of pharmaceutical- and food-grade ingredients in the e-liquids considerably reduce the likely presence of these phenolic compounds in e-cigarette aerosol.
Table 3 Phenolic compounds, carbonyls and ketones: per-puff concentration of e-cigarette vapour emissions, respective method air blanks and Ky1R6F cigarette smoke. Full size table
Carbonyls in cigarette smoke are formed mainly by pyrolysis of tobacco sugars83, whereas those in e-cigarettes are formed mainly by thermal degradation of PG and/or VG83,84,85. Flavourings may also contribute to the formation of carbonyls, as well as the characteristics of the e-cigarette devices, especially the applied voltage, coil resistance and wicking material47,48,49,86,87. Poor wicking efficiency may lead to a dry wick and overheated e-liquid (dry puff), which promotes the formation of carbonyls and other toxic compounds2,10,13,15. Coil location, orientation, resistance and wick material, as well as power output, have been shown to affect carbonyl generation significantly13,15,86. E-liquid physical properties are also important in carbonyl formation15,47,84,86. The viscosity and density of the e-liquid determine its mobility, capillary action, and delivery to the wick and coil, influencing the likelihood of a dry puff15.
Several studies have reported the presence of carbonyls in e-cigarette emissions at levels ranging from 0.07 to 413 µg puff–185,88,89. In our study, among the 14 analysed carbonyls (aldehydes and ketones), only five were quantifiable (acetaldehyde, acetone, formaldehyde, glyoxal and methylglyoxal) in the e-cigarette emissions at concentrations ranging from 0.02 to 0.19 µg puff–1 (Table 3). Of these, acetone was detected at the same level in e-cigarette emissions as the method air blank (0.04 µg puff–1). Detectable air blank values may arise from environmental contamination6,9,34,90. Acetaldehyde was quantified in BB57 emissions (0.10 µg puff–1) but was < LOQ in BB18 emissions, while formaldehyde was present in both (BB57, 0.04 µg puff–1; BB18, 0.07 µg puff–1). However, these two carbonyls were below the target levels proposed in the experimental voluntary standard published by the Association Française de Normalization (AFNOR; 16 µg puff–1 for acetaldehyde and 1 µg puff–1 for formaldehyde)91. Previous data indicate that the higher the percentage ratio of VG to PG, the higher the concentrations of carbonyl compounds emitted, especially acetaldehyde, acrolein and acetone84. In our study, both e-liquids had equivalent amounts of VG and PG; therefore, this ratio is likely to be irrelevant to the different concentrations of carbonyls detected in the two e-cigarette emissions, especially acetaldehyde. In a previous study, higher levels of acetaldehyde, acrolein and formaldehyde were generated in the emissions from an e-liquid without nicotine than in those from an e-liquid with nicotine; however, the observed carbonyl concentrations were strictly related to both the composition of the liquids and also the coil resistance47. In the presence of nicotine, the content of carbonyls, especially formaldehyde, was significantly higher with a 1.50-Ω coil than with a 0.25-Ω coil47. In our study, only acetaldehyde increased with the higher nicotine product (BB57); however, its concentration (0.10 µg puff–1) was still 160 times lower than the maximum level proposed by AFNOR (16 µg puff–1)91. A comparison of the emissions of an e-liquid with similar PG/VG ratio (1:1) emitted by a relatively similar Vype device (ePen) that uses a silica wick showed that formaldehyde at a concentration of 0.59 µg puff–1 was 8× higher and acetaldehyde at a concentration of 0.18 µg puff–1 was 2× higher than in the emissions presented herein (Table 3)92. In both studies, the values were below the maximum level proposed by AFNOR91.
From the studied carbonyls, only acetaldehyde, acrolein and formaldehyde are included in the nine WHO TobReg priority smoke toxicants55. Relative to cigarette smoke, their percentage of reduction in the e-cigarette emissions was ≥ 98.8%.
Among the 14 studied carbonyls, 7 were included in the new U.S. FDA HPHCs list for e-cigarettes53,54; namely, acetaldehyde, acrolein, formaldehyde and butyraldehyde, crotonaldehyde, and the diketones; diacetyl (2,3-butanedione) and acetyl propionyl (2,3-pentanedione). In the e-cigarette emissions, butyraldehyde and crotonaldehyde and acetyl propionyl were < LOD while acrolein and diacetyl were < LOQ. Acetoin, a precursor of diacetyl and acetyl propionyl, was also < LOD93.
Glyoxal and methylglyoxal are formed by thermal degradation or oxidation of PG and VG87. Glyoxal is considered mutagenic, while the related compound methylglyoxal has been identified as a metabolite during glycolysis and is thus naturally present in the body. Methylglyoxal is also present in foods and drinks such as honey and coffee. A lack of data has led to classification of methylgloxal as a Group 3 carcinogen (carcinogenicity to humans not classifiable) by IARC. Both compounds have been previously detected in e-cigarette emissions at concentrations of 0.07–0.94 and 0.09–33 µg puff–1, respectively86,88. In our study, glyoxal was present at lower levels (BB57 and BB18, 0.02 and 0.05 µg puff–1, respectively), while methylglyoxal was at concentrations of 0.13 and 0.19 µg puff–1 in BB57 and BB18, respectively (Table 3). Glyoxal was detected in the method air blank and therefore the actual levels in the e-cigarette emissions are potentially lower than reported in Table 3. Again assuming a worst-case exposure of 300 puffs day–1 spread over 8 h, the levels of glyoxal exposure to a consumer would still be more than 40 times lower than the occupational exposure limit of 0.10 mg m–377,94,95. The high standard deviation for glyoxal and methylglyoxal observed in BB18 e-cigarette emissions is probably related to an analytical sample matrix effect and/or batch variability83,96,97. Despite the high standard deviation, the percentage reduction of glyoxal and methylglyoxal in both e-cigarette emissions relative to cigarette smoke was ≥ 97.0% and ≥ 94.5%, respectively (Table 3).
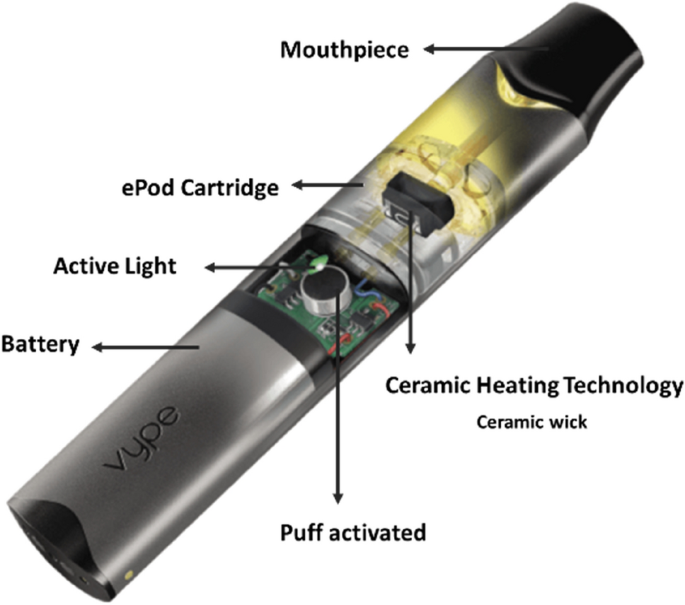
In our analysis, levels of carbonyls were considerably reduced relative both to other studies of e-cigarettes and to Ky1R6F cigarette smoke. Levels below the LOD or LOQ, or even below the threshold levels proposed by the AFNOR standard guidelines, provide evidence of the optimal operation conditions (e.g., adequate wick saturation without extreme coil heating) of the ceramic wick-based device.
Volatile organic compounds
Table 4 summarizes the levels of volatile organic compounds (VOCs) in the e-cigarette emissions and Ky1R6F cigarette smoke, along with the percentage reductions. Among the 23 VOCs analysed, levels were < LOD for both e-cigarette emissions, except for hydrogen cyanide (BB57, < LOQ), allyl alcohol (both < LOQ) and acetamide (BB57, < LOQ). Four of the VOCs, namely, acrylonitrile, benzene, propylene oxide and toluene, are listed by the U.S. FDA as compounds of concern for e-cigarettes53, while benzene and 1,3-butadiene are included in the nine WHO TobReg priority smoke toxicants55. The level of all of these compounds was < LOD with percentage reductions of ≥ 99.0% relative to Ky1R6F smoke (Table 4).
Table 4 Volatiles compounds: Per-puff concentration of e-cigarette vapour emissions, respective method air blanks and Ky1R6F cigarette smoke. Full size table
In particular, benzene, which may be formed by decarboxylation of benzoic acid, was undetectable in both e-cigarette emissions. Pankow et al.98 previously reported that benzene concentrations were largely undetectable for an e-cigarette with a single vertical coil and a cotton wick, but were more readily detected for a device with a single horizontal coil and a silica wick. Their results demonstrated the importance of the orientation of the coil and the type of wicking material in the formation of benzene. Our results showed that neither use of benzoic acid in the BB18 formulation nor the characteristics or operating conditions of the device contributed to benzene formation in the e-cigarette emissions. Pankow et al.98 also reported that benzene may be formed by the dehydration and cyclization of PG and VG, especially at high-power settings using a tank system with a single horizontal coil and a silica wick. Other studies have shown that 1,3-butadiene may be formed by VG degradation and is an important intermediate in the formation of benzene from VG84,98. Aromatic VOCs such as toluene, xylene, styrene and ethylbenzene may also be formed by thermal degradation of VG, where benzene plays an important role as an intermediate84. Benzene and other combustion-related compounds including acrylonitrile, isoprene and toluene may also be present in e-cigarette emissions as impurities of nicotine34. Percentage transfers to aerosol of ≥ 89% have been reported for these compounds after fortification of e-liquids at high levels (46–232 ng g–1)34. Other combustion-related compounds such as allyl alcohol and propylene oxide have been detected in e-cigarette emissions as thermal degradation products of PG and/or VG49,99. In our study, all these compounds were < LOD or < LOQ.
The above-cited studies show that the chemical composition of the e-liquid, the design of the device, and the temperature at which the e-liquids vaporize have a strong impact on the formation of VOCs and their transfer to e-cigarette emissions, especially those that originate primarily from heating of PG and VG. Our findings of levels < LOD or < LOQ in the e-cigarette emissions for the studied VOCs indicate the consistent supply of e-liquid by the ceramic wick without overheating of the coil and, consequently, overheating of the e-liquid. There was no considerable difference in levels of VOCs in the two emissions produced from e-liquids with different types of nicotine salt and different nicotine strengths. Moreover, there was a considerable reduction of VOCs in both e-cigarette emissions relative to cigarette smoke.
Flavouring compounds and acids
Next, we examined the e-cigarette emissions of flavouring compounds, together with acetic acid and propionic acid, as listed by the U.S. FDA as HPHCs of concern for e-cigarettes53,54. These compounds were not analysed in cigarette smoke because the Ky1R6F cigarette used in the study is an unflavoured US-blended cigarette and validated analytical methods for these compounds were not available.
All compounds were < LOD or < LOQ except for isoamyl acetate (isopentyl acetate) in the e-cigarette emissions (Table 5). This flavouring compound was used in both e-liquid formulations (BB57 and BB18), and therefore its presence in the e-cigarette emissions was expected. In a quantitative risk estimation performed in line with a published approach to the risk assessment of flavours in e-liquids100, the level of isoamyl acetate in the formulation was found to be supportable even if 100% of it were transferred to the aerosol. To further establish the level of risk, we again assumed a worst-case exposure of 300 puffs day–1 over 8 h77, which would result in isoamyl acetate exposure levels of 0.23 mg day–1 or 0.034 mg m–3. This is several orders of magnitude below various occupational exposure guidelines for isoamyl acetate, the lowest of which is 250 mg m–3, and below the acceptable daily intake of 3 mg kg–1 day–1 (180 mg day–1 for a 60-kg adult) established by the Joint FAO/WHO Expert Committee on Food Additives101,102.
Table 5 Flavouring compounds and acids: Per-puff concentration of e-cigarette vapour emissions and respective method air blanks. Full size table
Study limitations
The aim of this study was to undertake an assessment of the emissions generated by an e-cigarette using a ceramic wick-based technology and the comparison with conventional cigarette smoke. The generation of emissions from the e-cigarettes followed ISO 20768:2018 (55 mL puff volume, 3 s puff duration, 30 s puff frequency)52. ISO 20768:2018 lays out the essential requirements/conditions necessary to generate and collect e-cigarette emissions for analytical and comparison purposes in a robust and reproducible manner. The standard was developed building on the CORESTA (Cooperation Centre for Scientific Research Relative to Tobacco) recommended method no. 81 for machine puffing of e-cigarettes45,103. It is recognised that no single puffing regime can reflect the wide range of consumers’ puffing behaviour expected with e-cigarette use however, the use of ISO 20768:2018 is important for cross-product comparative purposes45. The application of different types of regime and its impact on the device performance is out of scope of this study. Nevertheless, the applied ISO standard puffing regime demonstrated that the emissions collected as block of 50 sequential puffs contained low levels of carbonyl compounds which would be associated with the thermal degradation of PG and VG. The carbonyl levels from the studied e-cigarettes emissions were either below the LOD or LOQ, or below the threshold levels proposed by the AFNOR standard guidelines, which provides evidence of the adequate wicking rate of the ceramic block without extreme coil heating under the test conditions. Whilst data generated from the first 50 puffs may not represent yields over the range of all puffs, especially as the liquid becomes depleted, the data are representative to enable generalised comparisons.