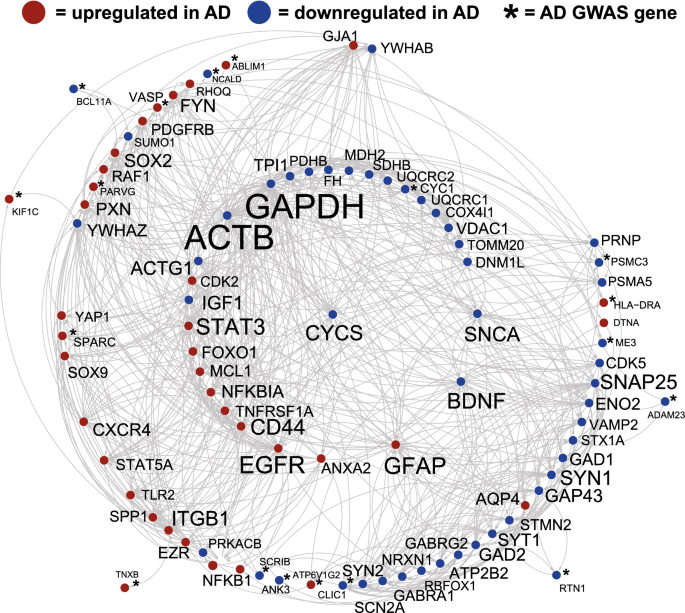
The AD portrait provides an overview of consistent gene expression dysregulation with AD across multiple brain regions and fits within the framework that similar pathologies are found across the CNS with AD1. An advantage of the AD portrait is that it allows for insights into global patterns, although it is less likely to reveal AD alterations that are specific to a region. However, given that many treatments have similar effects across brain regions, the portrait provides a platform for evaluating treatments, such as exercise, that could reverse the global patterns. As discussed below, the results of the AD portrait with treatments matches well those from individual AD datasets from specific regions with treatments, suggesting the portrait provides a robust platform for evaluating AD and potential treatments.
The top dysregulated gene was inositol-trisphosphate kinase, ITPKB (upregulated), that phosphorylates and converts the second messenger IP3 to IP4 and has been found to localize with actin32 and with amyloid plaques in human postmortem AD tissue33. While ITPKB is not commonly listed as being in the pathway for AD, its ability to affect IP3 levels that in turn influence intracellular calcium could connect it to the disease. Recent studies that support overexpression of ITPKB as causative of AD found in animal models that elevated ITBKB is linked to apoptosis and increased amyloid peptide production34 as well as TAU pathologies33,35. However, other studies suggest elevated ITPKB could be helpful in counteracting high Ca++ in mitochondria in neurologic pathologies36. Other inositol related genes dysregulated with AD were also upregulated, including ITPRID2, ITPRID, and ITPRIPL2A. The phospholipase C gene, PLCE1, that could affect IP3 levels was also upregulated. An understanding of whether or how elevated ITPKB contributes to AD remains to be determined. Interestingly, exercise reverses this overexpression pattern as discussed below.
The second most dysregulated gene was the astrocyte-specific gene, GFAP (upregulated), that is upregulated during inflammation and reactive gliosis, including within AD37,38,39. Other injury related genes were BDNF, AQP4, GAP43, GJA1, SOX9, CDK2, and EGFR. Overexpression of GFAP as occurs in Alexander's disease due to GFAP mutations, can lead to AD-like pathologies40. While early elevations of GFAP are expected as neuroprotective, prolonged overexpression could contribute to AD pathologies.
The Rho GTPase gene, RHOQ, was the third most dysregulated gene (upregulated) and it plays a role in actin cytoskeleton assembly41. Actin related processes were highly enriched among the top dysregulated AD genes. The transcription factor, NACC2 (upregulated), was the fourth most dysregulated gene and expression changes have been connected to Lewy bodies42. The dystrophin-related gene, DNTA (upregulated), was fifth ranked and DNTA localizes with the perivascular astrocytic endfoot and elevated levels are associated with increased AD pathologies43. Neuritin 1 (NRN1; downregulated) is involved in neuronal plasticity and associated with neurofibrillary tangles44 and was the sixth most altered gene in AD. The G protein regulating gene, RGS7 (downregulated) ranked as the seventh most disrupted gene and has been linked to multiple neuronal disorders45. While the majority of dysregulated genes are likely to have a contributory effect on AD, in future studies it will be important to evaluate which gene patterns are the most causative and which (if any) may seem like a dysregulation but are neuroprotective.
41 genes from a recent human GWAS AD study25 were among the top 1000 AD dysregulated genes (Fig. 1, Supplementary File 1), including the opioid neuropeptide, PNOC, downregulated and connected to AD46, the immune related gene, C4B, upregulated and linked with AD47, and the transcription factor, BCL11A, downregulated and associated with cortical neuron differentiation48. Additional GWAS genes previously linked with AD included: ANK3, MS4A6A, AGFG2, CYC1, HLA-DRA, MEG3, MT2A, NCALD, NEU1, PSMC3, SERPINB6, and SPARC (see full list in Supplementary File 1). How GWAS AD genes interact with or affect transcription of dysregulated AD genes remains to be determined.
In terms of common neurotransmitter pathways, the neuropeptides CRH, PNOC, VIP and SST (and its receptor SSTR1) were all downregulated as were the enzymes involves in GABA synthesis, GAD1 and GAD2. Potential roles for these signaling molecules in AD have been suggested46,49,50,51. Further, the GABA receptors GABBR2, GABRA1, GABRB3, GABRG2 and the glutamate receptors, GRIK1 and GRIK2 were also downregulated. While these neural signaling pathways were downregulated in AD, others were not suggesting potential specific roles for these signaling molecules in AD. As an example, elevation of GABA signaling has been proposed as a route for treatment of AD52.
Genes with the highest connections to one another (per STRING) within the top 1000 AD dysregulated genes included: ACTB, GAPDH, EGRF, SNAP25, STAT3, CYCS, SNCA, ACTG1, and BDNF (Fig. 1). It is possible that highly connected genes have an oversized effect on the AD phenotype given synergistic actions, but this is speculative. SNCA, GAPDH, and CYCS are part of the KEGG AD pathway53 and SNCA plays a role in the development of amyloid plaques54. Enrichment for the highly connected genes included mitochondrion, cell junction, immune system, actin cytoskeleton, and regulation of phosphorylation which is consistent with AD pathologies. BDNF is down regulated in the AD portrait and is part of both the BDNF and MAPK signaling pathways.
Forty-two transcription factors were identified with the top 1000 dysregulated AD genes also modified expression of other AD dysregulated genes (Supplementary File 2). STAT3, SOX9, ELK1, and SOX2 affected the most genes, but other transcription factors of interest included PAWR, GLIS3, AFF1, TCF3, FOXO1, BCL6, CEBPD, YAP1, RXRA, NFKB1, and NEUROD6 as these have previously been linked to AD within curated databases of diseases53,55,56,57. Transcription factors altered in AD that in turn affect expression of other AD genes could have a large effect on AD pathologies. Upregulated STAT3 contributes to astrogliosis and reversal of this pattern can mitigate AD phenotypes58. Similarly, SOX9 is elevated with AD and decreases in expression can reverse some AD markers59,60.
The female and male AD portraits were highly congruent with one another with 571 and 570 common genes the top 1000 up and downregulated genes, respectively. Further, no top 1000 gene was in the opposite direction. The RRHO heat map in Fig. 2 highlights the high congruence across levels of dysregulation. When focusing on the most dysregulated genes within each sex that were not found in the other sex, there were differences in highly interacting genes whereby in females GAPDH, CYCS, SOX2, and PHGDH had strong interactions, but in males TLR2, ITGB2, NFKB1, and CD53 interacted the most strongly. Our findings are similar to a recent study combining multiple datasets that also found significant overlap between males and females with AD when examining differential expression22. However, that study also found some sex differences in enrichment pathways and more extensive sex differences with AD when using Weighted Gene Co-expression Network Analysis (WGCNA)61 to identify gene networks. Our findings may differ for a few reasons, including that we did not systematically explore enrichment differences, we did not use WGCNA, and the results are based on different datasets. Given that the male AD portrait matched at high level the individual female AD datasets used to create the female AD portrait (and vice versa) (Supplementary File 1), we do not think our approach masked sex differences, but we cannot exclude this possibility.
Out of over 250 treatment datasets, the top three treatments were for exercise. A human CNS study comparing hippocampal gene expression in individuals with high versus low or high versus medium lifetime activity16 were the first and third top matches, respectively. The activity study also included a comparison of exercise modulated and AD modulated genes16 and identified some of the same genes of interest as in this study. The present approach differs in a few ways including that here multiple exercise/activity datasets were used, an exercise composite was created and used, and exercise was one of many possible treatments analyzed. The exercise composite that combined results from 11 exercise datasets, including those from human and rodents, was the second ranked treatment. In the top 20 were also two datasets examining exercise effects on the CNS in mice. Using UMAP as an alternative approach to identify the best treatments, exercise again stood out as a promising treatment (Fig. 7). These findings are consistent with a multitude of studies suggesting exercise in humans provides neuroprotective effects against development and progression of AD or related pathologies17,18,19,20,62,63,64.
The potential ability of exercise to reverse AD patterns was striking. For the first and second ranked treatments 409 and 344 AD genes were reversed while only 20 and 45 genes were in the same direction, respectively. Enrichment for AD genes reversed by the top exercise treatment included cell adhesion, cytoskeletal binding, neuron projection as well as multiple entries related to blood vessels, including blood vessel morphogenesis, circulatory system development, blood vessel development (Fig. 3B). These latter categories are of interest as decreased blood flow is associated with AD65 and exercise is posited to elevate brain blood flow as part of its effect on cognition66.
For the exercise composite (2nd ranked) enrichment of reversed AD genes included: transcription factor binding, actin binding, synapse organization, cell junction organization, brain development, BDNF signaling pathway, and a gene set from a recent study identifying interferon-stimulated network that is relevant to the innate immune response67. Previous studies have shown a link between interferon and exercise68 as well as possible roles for the innate immune system in the development of AD69. Whether or how exercise may invoke aspects of the innate immune response in the reversal of AD genes remains to be elucidated.
CDC42 (down in AD) is of interest as it had the greatest number of interactions with other genes reversed by exercise for the top two exercise datasets (Figs. 3, 5). CDC42 is a small GTPase of the Rho-subfamily and is connected to multiple pathways relevant to AD, including MAPK signaling, actin organization, cell junction, and CNS development. The possible role of CDC42 in AD may be complex as one line of research suggests inhibition as a pathway for treatment70, while another suggests activation as an approach to offset AD-like pathologies71. BDNF (down in AD) is reversed by the exercise composite and BDNF is well studied in terms of how it is upregulated by exercise and positively affects CNS function72,73 while its role in AD is still being explored74,75. Other genes reversed by exercise that are in the BDNF pathway include CDC42, NFKB1, MAPK8, and MAPK9. Twenty-two of the AD genes reversed by the exercise composite are part of the KEGG AD pathway, including SNCA, PSEN2, CALM3, GRIN2A, NFKB1, INSR, and TUBB. As indicated above, the top AD portrait dysregulated gene, ITPKB, is reversed by exercise. Together, exercise reverses a wide range of genes involved in a number of important AD-related processes. RRHO heat map analysis suggested exercise would also advantageously affect genes outside of the top 1000 dysregulated portrait genes (Figs. 4, 5).
The male and female portrait both had the same human exercise dataset as the top ranked treatment and the exercise composite ranked third in both. Further, the MetaVocano AD portrait as well as three recent meta-analysis datasets22,23,24 also had exercise as the top two treatments. While other AD meta-analysis studies exist76,77,78, full lists of genes were not provided so comparisons to treatments were not made. Importantly, exercise was also top ranked for both males and females within the two GEO datasets (GSE33000 and GSE44771)6,7 that were produced using the highest number of AD and control samples and came from prefrontal cortex and visual cortex, respectively. Results for 25 AD datasets that are brain region specific with each of the treatments are provided in Supplementary File 4. Exercise was highly ranked across the individual AD datasets with exercise ranked as the first and second best treatment for 13 of the 25 datasets. For another 8 datasets, one or more exercise treatments were in the top 10. There was not a clear pattern where tissue source affected matching to treatments and the best match to exercise (from hippocampus) was from an AD study in medial temporal cortex. The ROSMAP, Mayo, and MSBB studies each had exercise as a first or second top treatment, but interestingly there were high rankings for acetyl-L-carnitine which has been investigated for possible AD-related treatments79,80. In summary, the high ranking of exercise was robust across meta-analysis studies, across the individual datasets with the highest number of samples, and across most of the other AD datasets.
The antidepressant, fluoxetine, ranked fourth and a composite of fluoxetine that included results from 13 fluoxetine treatment datasets ranked 13th. In the top 25, there were four fluoxetine datasets plus the fluoxetine composite. Some of the top connected AD genes reversed by fluoxetine included BDNF, SYN1, VAMP2, GAD2, STX1A, STXBP1, and HDAC1 (Fig. 6). In both female and male AD portraits, the fluoxetine composite ranked 11th and 12th, respectively. This finding is consistent with recent work in animals and humans that fluoxetine can be a useful treatment for AD-related conditions81,82,83. When examining AD genes reversed by both fluoxetine and exercise composites, 44 genes were common, including BDNF, and a theoretical combining of the two treatments would reverse 549 AD genes. This finding supports recent work exploring the combination of both exercise and fluoxetine for AD and other disorders84.
Curcumin, the plant chemical from Turmeric, acting in cortex was ranked fifth and this is consistent with studies examining the therapeutic effects of curcumin in the treatment of AD85,86,87. However, curcumin acting in hippocampus had a slightly negative effect and datapoints from different ages had to be combined to achieve sufficient numbers. To our knowledge, a study examining curcumin effects in the CNS with a high number in each group has yet to be performed. Desipramine was in the top 10 and while animal studies indicate it can improve AD-related deficiencies88, the effects on cognition in humans with AD is less clear89. Safflower oil in a high fat diet was the 6th highest treatment, but the control in this study was flaxseed oil90, so whether the match is more due to one oil over the other is less clear. While D-serine matched as a possible treatment, the datasets for D-serine included an unusually high ratio of upregulated relative to downregulated genes overall91 and caution is needed when interpreting that result. The stimulant, cocaine, had three matches in the top 25 and while prolonged use of drugs of abuse induces clear cognitive deficits, the finding is consistent with studies exploring the ability of stimulants to mitigate some aspects of AD, such as apathy92. Overall, the ranking of treatments was similar for male and female portrait, although in males, curcumin was the second highest ranked treatment (File 1, Supplementary File 2). 25 individual AD datasets that are brain region specific were compared with each of the treatments and these can be explored in Supplementary File 4. Given that most treatments include only a few regions, comprehensive analysis that involves the same region is still limited. Future studies could focus more on data from specific brain regions (or cell types) from both AD datasets and treatments.
The two treatments that had the lowest treatment scores and were most similar to AD related to alcohol abuse and were from human datasets. Thus, these treatments could be viewed as risk factors for AD. The association of alcohol and AD is complex and still being evaluated93,94, but the findings are consistent with work suggesting alcohol abuse as a risk factor for AD95.
As a final step, the AD portrait was compared with a recent portrait of depression14 as comorbidity of depression and AD occurs in some individuals96,97. As shown in Fig. 8, there was a high matching of downregulated genes in both and these included BDNF, CRH, SST, GAD2, and PSEN2. The role of BDNF in depression is actively studied98,99 and a connection between exercise and increased BDNF as part of the antidepressant aspects of exercise have been evaluated100. In the depression portrait study, exercise also ranked as the top treatment14, but the extent of gene reversal was not nearly as large as for AD.
The treatments examined should be viewed as theoretical as the treatment expression studies varied widely across multiple factors, including sex, species (the majority of which were from rodents), numbers, brain region, treatment length, and platform. Most of the datasets were not created with the goal of understanding how the treatment might reverse AD dysregulation patterns and an understanding of experimental design is relevant for interpretation. Also, some recent proposed treatments for AD, such as aducanumab101, do not have corresponding large scale gene expression datasets, so they could not be included in this analysis.
We recently used a depression portrait to identify animal models with congruence to depression102 and ongoing useful steps could involve use of the AD portrait (or similar portraits) to evaluate and identify what animal model has the highest concordance with the AD brain signature. Advances in this area have already begun as a recent study identified mouse models that were congruent with coexpression modules found in AD24.
One goal for producing the AD portrait is to gain new insights into AD but also to produce a platform for identifying and evaluating new treatments at the large-scale gene expression level potential. As shown in Supplementary File 4, a final step involved modifying the AD portrait to include six datasets that came from three studies that were not in the original portrait. The two portraits are extremely similar and ITPKB and GFAP are the top two genes in both. With new datasets and integration approaches any AD portrait will always be ‘in progress’. With evolving portraits and individual datasets, a potentially promising approach is to identify multiple complementary treatments for AD, such as with exercise and fluoxetine.