Dietary fats may also have an effect on inflammation, which has a central role in the onset of many chronic pathologies. The first mechanism by which fats can affect inflammatory status is promotion of the translocation of microbial endotoxins, especially lipopolysaccharide (LPS), from the gut into the bloodstream [ 15 ]. LPS is a toxic cell-wall component of all our gut microbiome Gram-negative bacteria. The signaling of LPS is mediated through TLR4 receptors and leads to the stimulation of NF-kB, which in turn determines the secretions of many proinflammatory cytokines, such as IL-1, TNF-α, IL-6 and IL-8. SFAs seem to greatly stimulate inflammatory response because they are also structural components of LPS.
Although dietary SFAs are generally considered harmful to global health, these recent findings are quite controversial [ 9 ]. A possible explanation for these controversial results may derive from the type of SFA consumed in the diet. The absorption of short-chain fatty acids (SCFAs, two to six carbons) and medium-chain fatty acids (MCFAs, 8 to 12 carbons) occurs directly via portal circulation, while long-chain fatty acids (LCFAs, 14 to 20 or more carbons) are packaged into micelles and circulate via lymphatic-forming chylomicrons. While meat products are typically rich in LCFAs (i.e., palmitic and stearic acids), SCFAs (i.e., propionic acid and butyric acid) are produced when dietary fiber is fermented in the colon and are naturally present in milk and whole dairy products [ 10 ]. Scientific evidence from observational studies shows that higher intake of dairy products [ 11 ] and whole grains (rich in fiber) [ 12 ] are associated with a lower risk of CVD, while higher consumption of meat (red or processed) [ 13 ] is associated with higher risk. The evidence further suggests that the effects of SFA on lipid markers depend on the number of carbon atoms in the chain; a higher intake of lauric acid increases HDL cholesterol and reduces the TC to HDL ratio, while stearic, myristic and palmitic acids may raise LDL cholesterol [ 14 ].
Although calorie excess is the major determinant of weight gain, dietary fats have also been blamed for being the culprit in the dramatic increase in obesity and its associated diseases over the past half century. Over recent decades, dietary fat consumption has been discouraged but, in spite of the reduction in total daily individual fat intake by 10% and the increase in consumption of low-fat food, obesity rates are dramatically growing [ 5 ]. Furthermore, indistinctly cutting total fat intake from the diet inevitably leads to an increase in the consumption of highly processed grains and simple sugars while lowering the intake of liposoluble vitamins and unsaturated fats, especially from nuts, vegetable oils and fatty fish, which are particularly valuable for health [ 6 ].
Noncommunicable diseases (NCDs), such as cardio-metabolic disorders and cancer, are currently the main causes of global mortality, representing 71% of all deaths in the world [ 1 ]. Recent evidence shows that the major risk factor for these conditions is chronic subclinical low-grade inflammation [ 2 ], which is usually determined by physical inactivity, tobacco, pollution, sleep alterations, dysbiosis, and infections [ 3 ]. Among the triggers for inflammation, poor diet and excessive weight also play important roles [ 4 ]. The growth in the volume and number of adipocytes due to an excessive caloric intake leads to an increase in monocyte adhesion and recruitment to adipose tissue. Macrophages in adipocytes show many pro-inflammatory receptors, such as tumor necrosis factor receptors (TNFRs), Toll-like receptors (TLRs) and interleukin-1 receptor (IL-1R), as well as high activation of the nuclear factor-kB (NF-kB) transcription factors for pro-inflammatory cytokines [ 4 ]. Moreover, a condition of chronic low-grade inflammation may impair insulin sensitivity; insulin resistance worsens the inflammatory state, increasing abdominal obesity, intrahepatic fat stocking, vascular inflammation and endothelial dysfunction, which leads to an increase in cardiovascular risk [ 3 ].
Continuous variables are expressed as means and standard deviations (SDs), while categorical variables are expressed as frequencies of occurrence and percentages. Individuals were grouped by quartiles of total fat intake and distributions of background characteristics were compared between groups. Differences were tested with the chi-squared test for categorical variables, ANOVA for continuous variables distributed normally and the Kruskall–Wallis test for variables distributed non-normally. Energy-adjusted and multivariate logistic regression models were used to test the association between fat consumption and cardio-metabolic outcomes; the multivariate model adjusted for all other background characteristics (physical activity, educational status, occupational status, smoking status, alcohol consumption, menopausal status) was performed to test whether the observed associations were independent from the aforementioned variables. All reported p-values were based on two-sided tests and compared to a significance level of 5%. SPSS 17 (SPSS Inc., Chicago, IL, USA) software was used for all the statistical calculations.
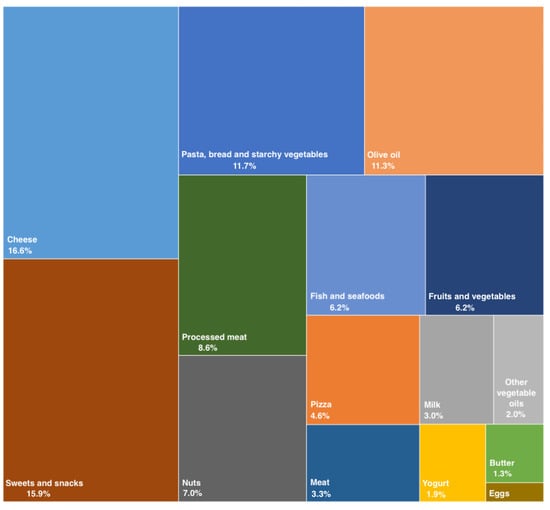
The nutritional assessment was conducted using two food frequency questionnaires (FFQs; a long and a short version) previously validated for the inhabitants of Sicily, south Italy [ 21 22 ]. The determination of the food ingested, the calories introduced and, especially, macro- and micro-nutrient intake were achieved through comparison with the food composition tables of the Research Center for Foods and Nutrition. The mean daily intake of each food was calculated in g or ml by considering the portion sizes provided in the FFQs and then converted to 24 h intake. Then, the content of total and specific fatty acids in each food was searched in the food composition tables of the Research Center for Foods and Nutrition and an estimation of their daily intake was calculated by multiplying the content of total and individual fatty acid molecules by the daily consumption of each food.
Anthropometric measurements were obtained following standard protocols [ 20 ]. Individuals were grouped according to body mass index (BMI) cut-offs as under/normal weight (BMI < 25 kg/m), overweight (from BMI 25 to 29.9 kg/m) and obese (BMI ≥ 30 kg/m). Arterial blood pressure was measured in sitting position and after at least 5 min of rest at the end of the physical examination. Due to the possibility of differences in blood pressure measurement, the measurements were taken three times from the right arm, relaxed and well-supported by a table, with an angle of 45° from the trunk. The mean of the last two measurements was considered for inclusion in the database. Patients were considered hypertensive when average systolic/diastolic blood pressure levels were equal to or more than 140/90 mmHg, in accordance with the European Society of Cardiology (ESC)/European Society of Hypertension (ESH) guidelines, or when participants had been previously diagnosed with hypertension. Patients were considered dyslipidemic or diabetic if diagnosed by a physician with hypercholesterolemia/hypertriglyceridemia or diabetes, respectively. Previous diagnosis of diseases was collected from the medical records of the referred general practitioner.
Data were collected with a presence-assisted interview through tablet computers. All subjects were supplied with a paper copy of the questionnaire in order to see each response option. However, the final answers were recorded instantly by the interviewer. The demographic information, such as age at recruitment, gender, highest educational level achieved, occupation (the most relevant job during the year before the investigation) or last occupation before retirement and marital status, were collected. Occupational status was classified as: (i) unemployed, (ii) low (unskilled workers), (iii) medium (partially skilled workers) and (iv) high (skilled workers). Educational status was categorized as: (i) low (primary/secondary), (ii) medium (high school) and (iii) high (university). The International Physical Activity Questionnaire (IPAQ) was used to examine motor activity [ 19 ], and it included a panel of questionnaires (five domains) investigating the time spent being physically active in the last week. In accordance with the IPAQ, physical activity level was classified as: (i) low, (ii) moderate and (iii) high. Smoking status was categorized as: (i) non-smoker, (ii) ex-smoker and (iii) current smoker, and alcohol consumption was classified as: (i) none, (ii) moderate drinker (0.1–12 g/d) and (iii) regular drinker (>12 g/d).
The Mediterranean Healthy Eating, Aging and Lifestyle (MEAL) study is an observational study aimed at examining the relationship between lifestyle and dietary habits in the Mediterranean region and NCDs. The original cohort included a sample of 2044 randomized adults (≥18 years old) from the main districts of Catania, a city in the south of Italy. The recruitment and data collection were carried out between 2014 and 2015. Details of the project protocol are published elsewhere. All subjects involved in the study were advised about the objective of the project and provided written informed consent. All the study methods were carried out following the Declaration of Helsinki (1989). The study protocol was examined and accepted by the relevant ethical committee.
The association between total and classes of dietary fats and metabolic outcomes is shown in Table 2 . Multivariate-adjusted analysis revealed a significant inverse association between total dietary fats and hypertension (for Q4, odds ratio (OR) = 0.57, 95% CI: 0.35, 0.91). Moreover, individuals reporting moderate consumption of total fats were less likely to have diabetes (for Q3, 0.27, 95% CI: 0.12, 0.61). Among single classes of dietary fats, individuals in the highest quartile of SFA intake were less likely to have hypertension (OR = 0.55, 95% CI: 0.34, 0.89). Moreover, subjects reporting moderate intake of MUFA were less likely to have hypertension and diabetes (OR = 0.61, 95% CI: 0.42, 0.88 and OR = 0.47, 95% CI: 0.22, 0.97, respectively). No associations were found between PUFA and any of the investigated outcomes.
4. Discussion
In the current study, we investigated the relationship between dietary fat subtype intake and cardio-metabolic risk factors in a cohort of Mediterranean adults. Interestingly, total SFA consumption was not detrimentally associated with any cardio-metabolic outcomes; conversely, individuals who had higher intake of total SFA were less likely to have hypertension and those who specifically consumed more SCSFA–MCSFA were less likely to have dyslipidemia and diabetes. Although SFAs have been assumed to be the main nutritional risk factor for cardio-metabolic diseases, recent studies have provided new controversial and interesting evidence suggesting that the SFA–CVD relationship may not be as strong as initially thought. A recent systematic review by the Cochrane group reported that a reduction in the intake of SFAs induced a 17% lowering of the risk of cardiovascular disease and that the beneficial effects increase when SFAs are replaced with PUFAs or starchy food [ 23 ]. However, cutting SFAs had a null effect on the other CVD end-points investigated and, furthermore, the putative adverse effect of SFAs on CVD events became non-significant when the analysis included only clinical trials that had successfully reduced SFA intake while removing those that were not successful [ 24 ]. The Prospective Urban Rural Epidemiological (PURE) study [ 25 ] conducted on 135,000 subjects without CVD demonstrated that increased consumption of total fats was linked with lower mortality and had a null association with CVD. Moreover, subjects in the highest quintile of SFA intake had a lower risk of stroke. Furthermore, a recent dose–response meta-analysis of cohort studies [ 8 ] highlighted that total fat, SFA, MUFA, and PUFA intake were not associated with CVD risk. Moreover, a meta-analysis of epidemiological studies conducted by SiriTarino [ 26 ] found no evidence that SFAs are associated with an increased risk of CHD. In addition, De Souza [ 27 ] and colleagues found no association between SFA intake and all-cause mortality, CHD, CHD mortality, ischemic stroke or type-2 diabetes among healthy subjects. A possible explanation for these controversial results on cardio-metabolic health may be the chain length of the fatty acids mainly consumed in diets [ 28 ]. SCFA may have a favorable impact on metabolism through activation of G protein-coupled receptors in endocrine and colon epithelial cells that, in turn, release anorectic hormones, such as glucagon-like peptide 1 (GLP) and peptide YY (PYY), which may contribute to reducing food intake and protecting individuals against obesity and diabetes, as shown in our results. Specifically, butyric acid (4:0) may have a beneficial effect on cardiovascular risk via inhibition of the NF-kB pathway and pro-inflammatory cytokines [ 29 ]. MCSFAs, such as caproic acid (6:0) and caprylic acid (8:0), have been shown in a cellular model to reduce the activity of fatty acid synthase (FAS), a primary enzyme of de novo lipogenesis that may contribute to the development of obesity and non-alcoholic fatty liver disease (NAFLD) [ 30 ]. Moreover, MCSFA may prevent endotoxic lipopolysaccharide (LPS)-mediated inflammation and lesions, which are linked to metabolic syndrome [ 31 ]. Caprylic acid (C8:0) and capric acid (C10:0) may reduce intestinal bile acid reabsorption with a simultaneous increase in excretion, which, in turn, lower TC and LDL-C [ 32 ].
In our cohort, myristic acid (14:0) intake was not associated with dyslipidemia and diabetes, while we found a positive association with hypertension. Although this molecule has been reported to play a role in post-translational protein changes and pathways that regulate several metabolic processes [ 33 ], data from the literature on its actual effect on metabolic health are not univocal: some studies reported potential beneficial effects from increasing HDL-C, such as reducing triglycerides levels, improving long-chain omega-3 levels in plasma phospholipids [ 34 ] and obesity-associated insulin resistance [ 35 ] and increasing LDL-C and apoB levels [ 36 ]. Among LCSFAs, in our study, stearic acid (18:0) was associated with a lower risk of diabetes and hypertension. Palmitic acid (16:0) and stearic acid (18:0) have been shown to increase cardiovascular risk through worsening of the lipid profile and alteration of inflammatory response [ 37 38 ], although the effects of stearic acid are less widely agreed on [ 39 ]. Moreover, stearic acid undergoes conversion to MUFA oleic acid (18:1) through the hepatic enzyme stearoyl–CoA–desaturase, which may in part explain the extensive detrimental effects.
n -3 and n -6 PUFAs could have contrasting roles in human health. Indeed, n -6 PUFAs, such as linoleic acid (LA, 18:2), were thought to have pro-inflammatory effects because they can be converted into arachidonic acid (AA, 20:4) with consequent production of pro-inflammatory eicosanoids and, at the same time, reduce the conversion of n -3 PUFA alpha-linolenic acid (ALA, 18:3) into eicosapentaenoic acid (EPA, 20:5) and/or docosahexaenoic acid (DHA, 22:6) by competing for the same enzymes [ n -6 PUFAs, but not n -3 PUFAs, may improve homeostasis model assessment—insulin resistance (HOMA-IR), lowering insulin in healthy subjects [ Despite the new nutritional scenario regarding the role of dietary fat in cardio-metabolic health, the American Heart Association recently remarked on the importance of limiting SFA and substituting it with PUFA to reduce cardiovascular risk [ 40 ]. The American Dietary Guidelines Advisory Committee showed strong evidence that replacing SFAs with PUFAs reduces the risk of CHD events and CVD mortality, but there is a paucity of evidence to establish whether this substitution affects the risk of stroke or heart failure [ 41 ]. Data from previous epidemiological studies [ 42 ] and clinical trials [ 43 ] have shown that replacing SFA with PUFA and MUFA reduces the risk of combined CVD events, mainly lowering LDL-C and reducing inflammation. However, a recent umbrella review showed that replacing SFA with PUFA does not convincingly reduce cardiovascular events or mortality, probably due to the original invalidity of the diet–heart hypothesis and numerous research biases [ 44 ]. In our study, neither total PUFA intake nor the individual PUFAs were associated with dyslipidemia. PUFAs have always been generally considered “healthy” fats. They are implicated in vascular function, cell membranes, the nervous system and inflammation, being precursors of eicosanoids and beneficial lipid mediators, such as resolvins, docosatrienes and protectins. Previous evidence suggested that-3 and-6 PUFAs could have contrasting roles in human health. Indeed,-6 PUFAs, such as linoleic acid (LA, 18:2), were thought to have pro-inflammatory effects because they can be converted into arachidonic acid (AA, 20:4) with consequent production of pro-inflammatory eicosanoids and, at the same time, reduce the conversion of-3 PUFA alpha-linolenic acid (ALA, 18:3) into eicosapentaenoic acid (EPA, 20:5) and/or docosahexaenoic acid (DHA, 22:6) by competing for the same enzymes [ 45 ]. This hypothesis has lost its strength based on the latest results from clinical studies on humans demonstrating that LA intake has little influence on conversion into AA, while, conversely, it may be converted into nitrosylated LA and 13-hydroxyoctadecadienoic acid, which show anti-inflammatory effects [ 46 ]. Interestingly, in our study, subjects reporting a higher intake of LA were less likely to be hypertensive in a linear way. The blood pressure-lowering effects of LA are probably due to changes in vasodilator prostaglandin metabolism [ 47 ]. Moreover, we found that the intake of EPA and DHA was inversely associated with hypertension. A recent dose–response meta-analysis showed that EPA and DHA may reduce the risk of CHD by lowering high blood pressure among people already diagnosed with hypertension [ 48 ]. These fatty acids may exert cardioprotective effects, mainly by reducing ventricular fibrillation, heart rate and platelet aggregation through various mechanisms, including improved synthesis of eicosanoids [ 49 50 ] and inhibition of the NF-kB pathway by acting on PPAR-gamma and certain G-protein-coupled receptors in macrophages and adipocytes, thus reducing the production of inflammatory cytokines [ 51 ]. Finally, PUFA may also influence the risk of type-2 diabetes by reducing insulin resistance, activating PPAR-alpha and suppressing sterol regulatory binding protein-1c (SREBP-1c) [ 52 ]. A recent meta-analysis of RCTs showed that substituting SFAs with PUFAs improved glycemia and insulin resistance [ 53 ]. However, in line with our results, not all subtypes of PUFA appear to have the same effects on type-2 diabetes risk. The latest data show that-6 PUFAs, but not-3 PUFAs, may improve homeostasis model assessment—insulin resistance (HOMA-IR), lowering insulin in healthy subjects [ 54 ]. Moreover, a recent study reported that high levels of LA, but not AR, are associated with a lower risk of becoming diabetic [ 55 ]. In our study, only subjects with the highest intake of LA were less likely to have diabetes while the association with other PUFA was null.
In addition to PUFAs, MUFAs have also been considered for replacing dietary SFAs, as highlighted by the latest American dietary guidelines, although evidence is weak. Although substitution of SFAs with MUFAs has been associated with a decrease in metabolic syndrome [ 56 ], findings from a meta-analysis of observational studies showed null results on risk of CVD [ 8 42 ]. Our results showed that individuals with higher intake of oleic acid (18:1) were less likely to have hypertension and diabetes, while no association was found for dyslipidemia. Typical sources of MUFAs in the Mediterranean region are nuts, which have been demonstrated to have potential benefits for metabolic disorders [ 57 ]. MUFAs may have anti-inflammatory and antioxidant properties, lowering endoplasmic reticulum stress, inhibiting NF-kB transcription factor and acting through AMP-activated protein kinase (AMPK) phosphorylation, and may also reduce the polarization of M1 macrophages to M2 macrophages [ 51 ]. However, recent published studies report a beneficial effect of extra virgin olive oil and, probably, its polyphenols rather than the simple intake of MUFAs [ 58 ]. When MUFAs were isocalorically replaced with a non-lipidic component, such as carbohydrates, observed effects on blood lipids were rather small or negligible, and a recent review even highlighted the potentially negative effects of olive oil and oleic acid if introduced in large quantities [ 59 ]. These findings are in line with those of our study concerning gadoleic acid (20:1), a long-chain MUFA that appeared to be detrimentally associated with each investigated outcome in our study. The Ludwigshafen Risk and Cardiovascular Health Study showed an inverse association of gadoleic acid with LDL-C, HDL-C and eGFR but direct correlations with markers of inflammation and endothelial activation, as well as heart failure [ 60 ]. In contrast, a randomized controlled trial found that the supplementation of LCMUFAs (gadoleic acid and cetoleic acid) improved endothelial function by lowering trimethylamine-N-oxide levels, IL-6 and TNF-α, possibly due to improved gut microbiota profile [ 61 ]. The debate on the effects of LCMUFAs (more than 18 carbons) remains inconclusive as research is still scarce.
The results of the current study should be considered in light of several limitations. First, the observational nature of this investigation did not make it possible to define a causal relation between variables but only an association. The cross-sectional design of the study may have limited the interpretation of the results, as they may have suffered from revere causation. Moreover, although we performed multivariate-adjusted logistic regression analyses, residual vulnerability to type-1 errors still exists and should be taken into account. Another limitation concerns the dietary assessment method: although there is a univocal and perfect approach to collecting dietary data (with integrated use of multiple 24 h recalls and dietary diaries being highly desirable), FFQs are known to potentially under- or overestimate food intake due to recall bias, portion size miscalculation and social desirability bias. Finally, cases were confirmed by medical visit, but potential undiagnosed patients may have led to inclusion of false-negative cases in the study sample. These findings require further investigation in studies that are better designed, more controlled and use a prospective approach.